PYQs With Solution
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections – A, B, C, D and E.
- Section A – Questions No. 1 to 20 are Multiple Choice Questions. Each question carries 1 mark.
- Section B – Questions No. 21 to 26 are Very Short Answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C – Questions No. 27 to 33 are Short Answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D – Questions No. 34 to 36 are Long Answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E – Questions No. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
SECTION-A (20 × 1 = 20)
Select and write the most appropriate option out of the four options given for each of the questions no. 1 to 20.
There is no negative marking for incorrect response.
1. Electrolysis of water is a decomposition reaction. The mass ratio
(MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is :
(A) 8 : 1
(B) 2 : 1
(C) 1 : 2
(D) 1 : 8
2. The products formed when Aluminium and Magnesium are burnt in the presence of air respectively are :
(A) Al₃O₄ and MgO₂
(B) Al₂O₃ and MgO
(C) Al₃O₄ and MgO
(D) Al₂O₃ and MgO₂
3. The following table shows the pH values of four solutions A, B, C and D on a pH scale:
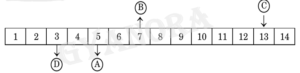
The solutions A, B, C and D respectively are of a
(A) Strong acid, weak acid, neutral, strong base
(B) Weak acid, neutral, weak base, strong base
(C) Weak acid, neutral, strong base, weak base
(D) Weak acid, neutral, strong base, strong acid
4. Consider the following reactions:
(i) Dilute hydrochloric acid reacts with sodium hydroxide.
(ii) Magnesium oxide reacts with dilute hydrochloric acid.
(iii) Carbon dioxide reacts with sodium hydroxide.
It is found that in each case:
(A) Salt and water are formed.
(B) Neutral salts are formed.
(C) Hydrogen gas is formed.
(D) Acidic salts are formed.
5. Reaction between two elements A and B forms a compound C. A loses electrons and B gains electrons. Which one of the following properties will not be shown by compound C ?
(A) It has high melting point.
(B) It is highly soluble in water.
(C) It has weak electrostatic forces of attraction between its oppositely charged ions.
(D) It conducts electricity in its molten state or aqueous solution.
6. The metals obtained from their molten chlorides by the process of electrolytic reduction are:
(A) Gold and silver
(B) Calcium and magnesium
(C) Aluminium and silver
(D) Sodium and iron
7. The formation of magnesium oxide is correctly shown in option:
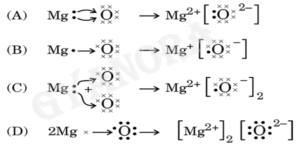
8. Secretion of less saliva in mouth will affect the conversion of :
(A) proteins into amino acids
(B) fats into fatty acids and glycerol
(C) starch into simple sugars
(D) sugars into alcohol
9. The plant hormone whose concentration stimulates the cells to grow longer on the side of the shoot which is away from light is :
(A) Cytokinins
(B) Gibberellins
(C) Adrenaline
(D) Auxins
10. The correct/true statement(s) for a bisexual flower is/are :
(i) They possess both stamen and pistil.
(ii) They possess either stamen or pistil.
(iii) They exhibit either self-pollination or cross-pollination.
(iv) They cannot produce fruits on their own.
(A) (i) only
(B) (iv) only
(C) (i) and (iii)
(D) (i) and (iv)
11. If pea plants with round and green seeds (RRyy) are crossed with pea plants having wrinkled and yellow seeds (rrYY), the seeds developed by the plants of F₁ generation will be :
(A) 50% round and green
(B) 75% wrinkled and green
(C) 100% round and yellow
(D) 75% wrinkled and yellow
12. The breakdown of glucose has taken the following pathway :
Glucose
→ Pyruvate + Energy
→ Lactic acid + Energy
The sites respectively are:
(A) Mitochondria and oxygen deficient muscle cells
(B) Cytoplasm and oxygen rich muscle cells
(C) Cytoplasm and yeast cells
(D) Cytoplasm and oxygen deficient muscle cells
13. Mirror ‘X’ is used to concentrate sunlight in solar furnace and mirror ‘Y’ is fitted on the side of the vehicle to see the traffic behind the driver. Which of the following statements are true for the two mirrors?
(i) The image formed by mirror ‘X’ is real, diminished and at its focus.
(ii) The image formed by mirror ‘Y’ is virtual, diminished and erect.
(iii) The image formed by mirror ‘X’ is virtual, diminished and erect.
(iv) The image formed by mirror ‘Y’ is real, diminished and at its focus.
(A) (i) and (ii)
(B) (ii) and (iii)
(C) (iii) and (iv)
(D) (i) and (iv)
14. An old person is suffering from an eye defect caused by weakening of ciliary muscles and diminishing flexibility of the eye lens. If the defect of vision is ‘a’ which can be corrected by lens ‘b’, then ‘a’ and ‘b’ respectively are :
(A) Hypermetropia and convex lens
(B) Presbyopia and bifocal lens
(C) Myopia and concave lens
(D) Myopia and bifocal lens
15. Which of the following groups do not constitute a food chain ?
(i) Wolf, rabbit, grass, lion
(ii) Plankton, man, grasshopper, fish
(iii) Hawk, grass, snake, grasshopper, frog
(iv) Grass, snake, wolf, tiger
(A) (i) and (iv)
(B) (i) and (iii)
(C) (ii) and (iii)
(D) (ii) and (iv)
16. The percentage of solar energy which is not converted into food energy by the leaves of green plants in a terrestrial ecosystem is about:
(A) 1%
(B) 10%
(C) 90%
(D) 99%
For Questions 17 to 20
Two statements are given — one labelled as Assertion (A) and the other labelled as Reason (R).
Select the correct answer from the codes given below:
(A) Both A and R are true and R is the correct explanation of A.
(B) Both A and R are true, but R is not the correct explanation of A.
(C) A is true, but R is false.
(D) A is false, but R is true.
17.
Assertion (A) : Decomposition reactions are generally endothermic reactions.
Reason (R) : Decomposition of organic matter into compost is an exothermic process.
18.
Assertion (A) : A human child bears all the basic features of human beings.
Reason (R) : It looks exactly like its parents, showing very little variations.
19.
Assertion (A) : No two magnetic field lines are found to cross each other.
Reason (R) : The compass needle cannot point towards two directions at the point of intersection of two magnetic field lines.
20.
Assertion (A) : The amount of ozone in the atmosphere began to drop sharply in the 1980s.
Reason (R) : The oxygen atoms combine with molecular oxygen to form ozone.
ANSWERS OF SECTION-A
- (D)
- (B)
- (B)
- (A)
- (C)
- (B)
- (B)
- (C)
- (D)
- (C)
- (C)
- (D)
- (A)
- (B)
- (A)
- (D)
- (D)
- (C)
- (A)
- (B)
SECTION-B (2 x 6 = 12)
Questions no. 21 to 26 are Very Short Answer Type questions.
Each question carries 2 marks.
21. A student performs the following experiment in his school laboratory.
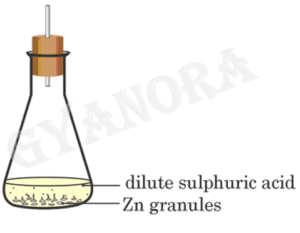
List two observations to justify that in this experiment a chemical change has taken place.
22. Draw labelled diagrams to show different stages of budding in Hydra.
23.
(a) Besides minimising the loss of blood, why is it essential to plug any leak in a blood vessel? Name the component of blood which helps in this process and state how this component performs this function.
OR
(b)
(i) The transport system in plants is relatively slower than in animals. Give reasons.
(ii) State the role of phloem in the transport of materials in plants.
24. An object is placed at a distance of 60 cm from a concave lens of focal length 30 cm. Use lens formula to find the position of the image formed in this case.
25.
(a) A wire of resistance R is cut into three equal parts. If these three parts are then joined in parallel, calculate the total resistance of the combination so formed.
OR
(b) Define electric power. When do we say that the power consumed in an electric circuit is 1 watt ?
26. “Excessive use of chemicals and pesticides in agriculture adversely affect the environment.” Justify this statement.
ANSWERS OF SECTION-B
21. The given experiment shows that a chemical change has taken place because a new substance is formed during the reaction. Also, the change is irreversible in nature, which is a characteristic feature of a chemical change.
22.
In Hydra, reproduction takes place by budding. First, a small outgrowth called a bud appears on the parent body. The bud gradually grows and develops tentacles. Finally, the fully formed bud detaches itself from the parent body and becomes an independent organism.
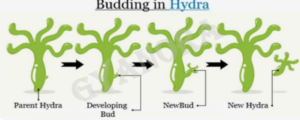
Stages of budding in Hydra:
- Bud formation
- Growth of bud
- Detachment of bud
23. (a)
Besides preventing loss of blood, plugging a blood vessel is essential to stop the entry of disease-causing microorganisms into the body. Platelets help in this process by forming a clot at the site of injury, which seals the damaged blood vessel.
OR
23. (b)
The transport system in plants is slower than in animals because plants do not have a pumping organ like the heart and transport mainly occurs by diffusion. Phloem helps in transporting food prepared in the leaves to all parts of the plant.
24.
Given,
Object distance (u) = –60 cm
Focal length (f) = –30 cm
Using lens formula,
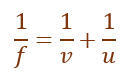
Substituting the values,
![]()
On solving,
v = –20 cm
Thus, the image is formed at a distance of 20 cm in front of the lens.
25. (a)
When a wire of resistance R is cut into three equal parts, the resistance of each part becomes R/3. When these three parts are connected in parallel, the equivalent resistance of the combination becomes R/9.
OR
25. (b)
Electric power is defined as the rate at which electrical energy is consumed in a circuit. The power consumed in an electric circuit is said to be 1 watt when 1 joule of electrical energy is consumed in 1 second.
26.
Excessive use of chemicals and pesticides in agriculture causes soil and water pollution. These harmful substances enter the food chain and affect human health. They also destroy useful microorganisms present in the soil, thereby reducing soil fertility.
SECTION-C (3 x 7 =21)
Questions no. 27 to 33 are Short Answer Type questions.
Each question carries 3 marks.
27.
(a) “Displacement reactions also play a key role in extracting metals in the middle of the reactivity series.”
Justify this statement with two examples.
(b) Why can metals high up in the reactivity series not be obtained by reduction of their oxides by carbon ?
28.
(a) With the help of an activity, explain the conditions under which iron articles get rusted.
OR
(b)
(i) Name two metals which react violently with cold water. List any three observations which a student notes when these metals are dropped in a beaker containing water.
(ii) Write a test to identify the gas evolved (if any) during the reaction of these metals with water.
29. Plants have neither a nervous system nor muscles, even then they respond to stimuli. For example, the leaves of chhui-mui (touch-me-not) plant when touched begin to fold up and droop.
(a) How is the information communicated in “touch-me-not” plants ?
(b) What enables the plant cells to bring out the observable response ?
(c) Differentiate the movement mentioned above from the movement of tendrils in a pea plant.
30.
(a) What are chromosomes ?
(b) Explain in brief how stability of DNA content of a species is ensured in sexually reproducing organisms.
31. Draw ray diagrams to show the nature, position and relative size of the image formed by a convex mirror when the object is placed
(i) at infinity and
(ii) between infinity and pole P of the mirror.
32. Consider the following electric circuit:
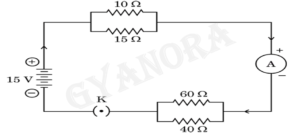
Calculate the values of the following:
(a) The total resistance of the circuit
(b) The total current drawn from the source
(c) Potential difference across the parallel combination of 10 Ω and 15 Ω resistors
33.
(a) Write the relationship between resistivity and resistance of a cylindrical conductor of length l and area of cross-section A. Hence derive the SI unit of resistivity.
(b) Why are alloys used in electrical heating devices?
ANSWERS OF SECTION-C
27. (a)
Displacement reactions play an important role in the extraction of metals placed in the middle of the reactivity series. In these reactions, a more reactive metal displaces a less reactive metal from its compound. For example, iron displaces copper from copper sulphate solution and zinc displaces copper from copper sulphate. Thus, such reactions help in obtaining metals from their compounds.
(b)
Metals placed high up in the reactivity series cannot be obtained by reduction of their oxides using carbon because these metals are more reactive than carbon. Carbon is unable to remove oxygen from their oxides due to their strong affinity for oxygen.
28. (a)
Iron articles get rusted in the presence of both oxygen and moisture. This can be explained by an activity in which iron nails are placed in different test tubes containing dry air, water and salt water. It is observed that rusting occurs fastest in salt water, slower in water and does not occur in dry air. This shows that both air and moisture are essential for rusting.
OR
28. (b)
Sodium and potassium are two metals that react violently with cold water. When these metals are dropped in water, vigorous reaction takes place, hydrogen gas is evolved and a lot of heat is produced. The gas evolved is identified by bringing a burning matchstick near it, which burns with a pop sound, confirming the presence of hydrogen gas.
29.
In touch-me-not plants, information is communicated through chemical signals instead of nerves. The observable response is brought about by a change in the turgor pressure of plant cells. The movement of leaves in touch-me-not is non-directional, whereas the movement of tendrils in pea plants is directional and depends on the direction of the stimulus.
30.
Chromosomes are thread-like structures present in the nucleus of a cell and they carry genes, which contain hereditary information. In sexually reproducing organisms, stability of DNA content is ensured because meiosis produces gametes with half the chromosome number and fertilisation restores the original number in the offspring.
31.
When an object is placed at infinity in front of a convex mirror, the image is formed at the focus behind the mirror and is highly diminished and virtual. When the object is placed between infinity and the pole of the mirror, the image is formed between the focus and pole, remains virtual, erect and diminished in size
32.
First, the total resistance of the circuit is calculated by combining the resistances using the appropriate formula. Then, the total current drawn from the source is calculated using Ohm’s law. Finally, the potential difference across the parallel combination of the 10 Ω and 15 Ω resistors is determined using the relation V = IR.
33. (a)
The resistance of a cylindrical conductor is related to its resistivity by the relation
R = ρl/A,
where l is the length and A is the area of cross-section. The SI unit of resistivity is ohm metre (Ω m).
(b)
Alloys are used in electrical heating devices because they have high resistivity and high melting point, due to which they do not burn easily when heated.
SECTION-D (3 x 5 = 15)
Questions no. 34 to 36 are Long Answer Type questions.
Each question carries 5 marks.
34.
(a)
(i) Draw two isomeric structures of Butene (C₄H₈).
(ii) Name the following compounds:
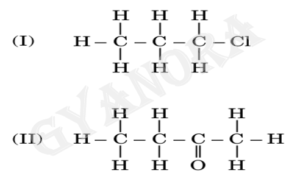
(iii) Write the chemical equations for the following reactions.
Mention one essential condition each for these reactions to take place.
(I) Ethanol undergoes complete oxidation
(II) Propene undergoes hydrogenation
(III) Ethanoic acid reacts with ethanol
OR
(b)
(i) A carbon compound X is a good solvent. On reaction with sodium, X forms two products Y and Z. Z is used to convert vegetable oil into vegetable ghee. Identify and name X, Y and Z. Also write the equation of reaction of X with sodium to justify your answer.
(ii) Write chemical equations to show what happens when ethanol :
(I) burns in oxygen/air.
(II) is heated at 443 K in excess concentrated H₂SO₄.
(III) reacts with acidified potassium dichromate.
35.
(a)
(i) Write the functions of the following parts of human female reproductive system :
(I) Ovary
(II) Fallopian tube
(III) Uterus
(ii) State briefly two contraceptive methods used by human males.
OR
(b)
(i) Differentiate between self-pollination and cross-pollination.
(ii) Identify A, B and C in the diagram given below and write one function of each.
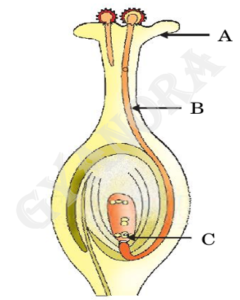
36.
(a)
(i) The power of a lens ‘X’ is –2.5 D. Name the lens and determine its focal length in cm. For which eye defect of vision will an optician prescribe this type of lens as a corrective lens ?
(ii) “The value of magnification ‘m’ for a lens is –2.” Using new Cartesian Sign Convention and considering that an object is placed at a distance of 20 cm from the optical centre of this lens, state:
(I) the nature of the image formed;
(II) size of the image compared to the size of the object;
(III) position of the image; and
(IV) sign of the height of the image.
(iii) The numerical values of the focal lengths of two lenses A and B are 10 cm and 20 cm respectively. Which one of the two will show higher degree of convergence/divergence ? Give reason to justify your answer.
OR
(b)
(i) Draw a ray diagram to show the refraction of a ray of light through a rectangular glass slab when it falls obliquely from air into glass.
(ii) State Snell’s law of refraction of light.
(iii) Differentiate between the virtual images formed by a convex lens and a concave lens on the basis of:
(I) object distance, and
(II) magnification.
ANSWERS OF SECTION-D
34. (a)
(i) The two isomeric structures of Butene (C₄H₈) are But-1-ene and But-2-ene. These compounds have the same molecular formula but different structural arrangements, hence they are called isomers.
(ii)
(I) The given compound is Butane.
(II) The given compound is Butan-1-ol.
(iii)
(I) When ethanol undergoes complete oxidation, it forms carbon dioxide and water in the presence of an oxidising agent.
![]()
(II) When propene undergoes hydrogenation in the presence of a nickel catalyst, it forms propane.
![]()
(III) When ethanoic acid reacts with ethanol in the presence of concentrated sulphuric acid, ethyl ethanoate is formed.
![]()
OR
34. (b)
The carbon compound X is ethanol, which is a good solvent. When ethanol reacts with sodium, it forms sodium ethoxide (Y) and hydrogen gas (Z). Hydrogen gas is used for converting vegetable oil into vegetable ghee.
![]()
When ethanol burns in air, it produces carbon dioxide and water. When heated at 443 K with excess concentrated sulphuric acid, ethanol undergoes dehydration to form ethene. When ethanol reacts with acidified potassium dichromate, it is oxidised to ethanoic acid.
35. (a)
The ovary produces female gametes called ova and also secretes female hormones. The fallopian tube carries the ovum from the ovary to the uterus and is the site of fertilisation. The uterus provides nourishment and a suitable environment for the development of the embryo.
Two commonly used contraceptive methods by human males are the use of condoms and vasectomy. Condoms prevent the entry of sperms into the female reproductive system, while vasectomy involves cutting and tying of the vas deferens.
OR
35. (b)
Self-pollination occurs when pollen grains are transferred from the anther to the stigma of the same flower, whereas cross-pollination occurs when pollen grains are transferred from one flower to another flower of a different plant of the same species.
In the given diagram, A, B and C represent different parts of the flower. Each part performs a specific function such as protection of the flower, production of pollen grains and formation of seeds after fertilisation
36. (a)
The lens X is a concave lens because its power is negative. Its focal length is –40 cm. This type of lens is used to correct myopia (short-sightedness).
When the magnification of a lens is –2, the image formed is real and inverted and its size is twice that of the object. Since the object is placed at a distance of 20 cm, the image is formed at a distance of 40 cm from the lens on the opposite side. The height of the image is taken as negative according to sign convention.
Between the two lenses A and B, lens A with focal length 10 cm shows a higher degree of convergence because a lens with a smaller focal length bends light rays more strongly.
OR
36. (b)
When a ray of light passes obliquely from air into a rectangular glass slab, it bends towards the normal at the first surface and away from the normal at the second surface, emerging parallel to the incident ray.
Snell’s law of refraction states that the ratio of the sine of the angle of incidence to the sine of the angle of refraction is constant for a given pair of media.
A convex lens forms a virtual image only when the object is placed between the optical centre and focus, whereas a concave lens always forms a virtual image. The magnification produced by a convex lens can be greater than one, while that produced by a concave lens is always less than one.
SECTION-E (3 x 4 = 12)
The following questions are Source-based/Case-based questions.
Read the case carefully and answer the questions that follow.
37. Seawater contains many salts dissolved in it. Common salt is separated from these salts. Deposits of solid salt are also found in several parts of the world. These large crystals are often brown due to impurities. This is called rock salt and is mined like coal. The common salt is an important raw material for chemicals of daily use.
(a) Write balanced chemical equations to show the products formed during electrolysis of brine. (1)
(b) List two uses of any one product obtained during electrolysis of brine. (1)
(c)
(i) A mild non-corrosive basic salt ‘A’, used for faster cooking, is strongly heated to produce a compound ‘B’, that is used for removing permanent hardness of water. Identify A and B and also write the equation for the reaction that occurs when A is heated. (2)
OR
(c)
(ii) Define water of crystallisation. Give two examples of salts that have water of crystallisation. (2)
38. The maintenance functions of all living organisms must go on even when they are not doing anything particular. Even when we are just sitting in a class or even asleep, this maintenance job has to go on. These maintenance processes require energy to prevent damage and break-down of cells and tissues, which is obtained by the individual organism from the food prepared by the autotrophs, called producers.
(a) Name and define the process by which green plants prepare food. (1)
(b) Write chemical equation involved in the above process. (1)
(c)
(i) State in proper sequence the events that occur in synthesis of food by desert plants. (2)
OR
(c)
(ii) Explain giving reasons what happens to the rate at which the green plants will prepare food (2)
(I) during cloudy weather, and
(II) when stomata get blocked due to dust.
39. In our homes, we receive the supply of electric power through a main supply also called mains, either supported through overhead electric poles or by underground cables. In our country the potential difference between the two wires (live wire and neutral wire) of this supply is 220 V.
(a) Write the colours of the insulation covers of the line wires through which supply comes to our homes. (1)
(b) What should be the current rating of the electric circuit (220 V) so that an electric iron of 1 kW power rating can be operated ? (1)
(c)
(i) What is the function of the earth wire ? State the advantage of the earth wire in domestic electric appliances such as electric iron. (2)
OR
(c)
(ii) List two precautions to be taken to avoid electrical accidents. State how these precautions prevent possible damage to the circuit/appliance. (2)
ANSWERS OF SECTION-E
37.
(a) The chemical reaction involved in the electrolysis of brine is:
![]()
This process is known as the chlor-alkali process.
(b) Sodium hydroxide is used in the manufacture of soaps and detergents and also in paper industries. Chlorine gas is used for disinfecting drinking water and in the manufacture of bleaching powder.
(c) Substance A is baking soda (sodium hydrogen carbonate) and substance B is washing soda (sodium carbonate).
38.
(a) The process responsible for the conversion of solar energy into chemical energy in plants is photosynthesis.
(b) The balanced chemical equation for photosynthesis is:
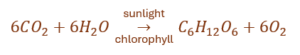
(c) On a cloudy day, the rate of photosynthesis decreases because sunlight is less available. Dust on the surface of leaves blocks sunlight and stomata, thereby reducing the rate of photosynthesis.
39.
(a) The colour coding of domestic electric wires is such that the live wire is red and the neutral wire is black.
(b) Given,
Power of heater = 1000 W
Voltage = 220 V
Using the formula,
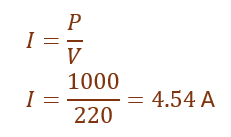
Thus, the current drawn by the heater is approximately 4.5 A.
(c) An earth wire is used as a safety measure to prevent electric shock. It provides a low-resistance path for excess current to flow into the ground, thereby protecting the user from electric shock in case of leakage of current.
x——————x—————-x——————x
Revise your Science Concepts for Board Exam by CLICK HERE or visit Gyanora.in
Practice Chapter-Wise Extra Questions of Science for Your Board Exams by CLICK HERE
OR Explore other Chapter’s Extra Questions:
Chapter-2 Acid, Bases, and Salt
Chapter-3 Metals and Non-Metals
Chapter-4 Carbon and it’s Compounds
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6

