PYQ Paper 2025
General Instructions for this Paper:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections – A, B, C, D and E.
- Section A – Questions No. 1 to 20 are Multiple Choice Questions. Each question carries 1 mark.
- Section B – Questions No. 21 to 26 are Very Short Answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C – Questions No. 27 to 33 are Short Answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D – Questions No. 34 to 36 are Long Answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E – Questions No. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
SECTION A (20 × 1 = 20)
Questions No. 1 to 20 are Multiple Choice Questions. Only one of the choices is correct. Select and write the most appropriate option out of the four options given for each.
1. Consider the following chemical equation:
p Al + q H₂O ⎯→⎯ r Al₂O₃ + s H₂
To balance this chemical equation, the values of ‘p’, ‘q’, ‘r’ and ‘s’ must be respectively:
(A) 3, 2, 2, 1
(B) 2, 3, 3, 1
(C) 2, 3, 1, 3
(D) 3, 1, 2, 2
2. Which of the given option represents a family of salts ?
(A) NaCl, Na₂SO₄, CaSO₄
(B) K₂SO₄, Na₂SO₄, CaSO₄
(C) NaNO₃, CaCO₃, Na₂CO₃
(D) MgSO₄, CuSO₄, MgCl₂
3. The most common method of extraction of metals from their oxide ores is:
(A) Reduction with carbon
(B) Reduction with hydrogen
(C) Reduction with aluminium
(D) Electrolytic reduction
4. Given below are the structures of some hydrocarbons. Select the two structures which are related to each other from the given options:
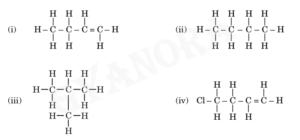
(A) (i) and (iv)
(B) (ii) and (iv)
(C) (ii) and (iii)
(D) (i) and (iii)
5. Choose the incorrect statement about the common reaction used in hydrogenation of vegetable oils.
(A) It is an addition reaction.
(B) It takes place in the presence of nickel or palladium catalyst.
(C) The product contains only single bonds between carbon atoms.
(D) It is an addition reaction which occurs in the presence of an acid
catalyst.
6. Match Column-I with Column-II and select the correct option from the choices provided.
| Column-I | Column-II |
| a. Site of fertilisation of egg with the sperm | (i) Vagina |
| b. Site of implantation of embryo | (ii) Uterus |
| c. Site of entry of sperm into the female reproductive tract | (iii) Oviduct |
| d. Site through which the waste materials generated by the developing embryo are removed | (iv) Placenta |
| (v) Cervix |
(A) a-(ii), b-(i), c-(v), d-(iv)
(B) a-(iii), b-(i), c-(v), d-(iv)
(C) a-(iv), b-(ii), c-(iii), d-(i)
(D) a-(iii), b-(ii), c-(i), d-(iv)
7. The part of the brain which maintains the posture and balance of the body is:
(A) Pons
(B) Cerebrum
(C) Cerebellum
(D) Medulla
8. The plant hormone present in greater concentration in the areas of rapidly dividing cells is :
(A) Auxin
(B) Cytokinins
(C) Gibberellins
(D) Abscisic acid
9. Select a pair of bisexual flowers from the following :
(A) Papaya and mustard
(B) Hibiscus and mustard
(C) Hibiscus and papaya
(D) Hibiscus and watermelon
10. The gastric glands present in the wall of the stomach release :
(A) Mucus and Trypsin
(B) Pepsin and Trypsin
(C) Mucus and Pepsin
(D) Pepsin and Salivary amylase
11. Absolute refractive index of water and glass is 4/3 and 3/2 respectively. If the speed of light in glass is 2 × 10⁸ m/s, the speed of light in water is :
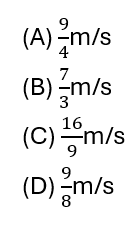
12. When a beam of white light passes through a region of very fine dust particles, the colour of light that scatters the most in that region is :
(A) Red
(B) Orange
(C) Blue
(D) Yellow
13. A wire of length ‘l’ is gradually stretched so that its length increases to 3l. If its original resistance is R, then its new resistance will be :
(A) 3R
(B) 6R
(C) 9R
(D) 27R
14. Which one of the following statements is not true about a bar magnet ?
(A) It sets itself in north-south direction when suspended freely.
(B) It has attractive power for iron filings.
(C) It produces magnetic field lines.
(D) The direction of magnetic field lines inside a bar magnet is from its
north pole to its south pole.
15. The strength of magnetic field produced inside a long straight current carrying solenoid does not depend upon :
(A) Number of turns in the solenoid
(B) Direction of current flowing through the solenoid
(C) Material of the core filled inside the solenoid
(D) Radius of the coil of the solenoid
16. Other than the abiotic components, which of the given biotic components are not required to make an aquarium with small herbivorous fishes a self-sustaining system ?
(i) Aquatic plants and aquatic animals
(ii) Terrestrial plants and terrestrial animals
(iii) Decomposers as bacteria and fungi
(iv) Consumers as clown fishes and sea urchins
(A) (i) and (iv)
(B) (ii) and (iii)
(C) (i) and (iii)
(D) (ii) and (iv)
For Questions number 17 to 20, two statements are given — one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer from the codes (A), (B), (C) and (D).
17. Assertion (A): Hydrogen gas is not evolved when a metal reacts with nitric acid.
Reason (R): Nitric acid is a strong reducing agent and reduces the hydrogen produced in the reaction to water.
18. Assertion (A): In our actions of writing or talking, our nervous system communicates with the muscles.
Reason (R): Cranial nerves and spinal nerves form the peripheral nervous system.
19. Assertion (A): Magnetic field lines around a bar magnet never intersect each other.
Reason (R): Magnetic field produced by a bar magnet is a quantity that has both magnitude and direction.
20. Assertion (A): Use of jute bags for shopping reduces pollution.
Reason (R): Jute is biodegradable and its bag may be reused as and when needed.
ANSWERS OF SECTION A
- (B)
- (A)
- (D)
- (A)
- (A)
- (A)
- (C)
- (B)
- (B)
- (D)
- (A)
- (A)
- (C)
- (B)
- (D)
- (A)
- (D)
- (A)
- (B)
- (A)
SECTION B (2 x 6 = 12)
Questions no. 21 to 26 are Very Short Answer Type questions.
21.
(a) List the possible sources of energy required in decomposition reactions. Illustrate any one with a suitable example.
OR
(b) What is observed when hydrated ferrous sulphate crystals are heated in a dry boiling tube ? Give balanced chemical equation(s) of the reactions(s) that occur(s).
22.
(a) Write the formula of the ions which (i) acids, and (ii) bases generate in water solutions.
(b) Dry HCl gas does not change the colour of dry litmus paper. Why ?
23.
State the main function of veins in human circulatory system. Why do they not need thick walls ?
24.
(a) Explain how the proteins control the ‘characteristics’ in an organism with the help of an example of ‘tallness’ trait in pea plant.
(b) Name the section of DNA that controls the ‘characteristics’ in an organism.
25.
(a) A student has difficulty in reading his textbooks but can read the blackboard clearly while sitting in the last row. Name the defect of vision the student is suffering from. List two reasons due to which this defect arises. Write the nature of the lenses required to correct this defect.
OR
(b) Draw a ray diagram to show the path of a ray of light falling obliquely on one of the refracting faces of a triangular glass prism and mark the angle of deviation on it.
26.
An electric kettle is rated 230 V; 1000 W. Calculate the resistance of its heating element when in operation.
ANSWERS OF SECTION B
21. (a)
Decomposition reactions require energy to break a compound into simpler substances. The possible sources of energy are heat, light and electricity.
For example, when calcium carbonate is heated, it decomposes into calcium oxide and carbon dioxide.
CaCO₃ (s) ⟶ CaO (s) + CO₂ (g)
OR
21. (b)
When hydrated ferrous sulphate crystals are heated in a dry boiling tube, they first lose water of crystallisation and change their colour from green to white. On further heating, it decomposes to form ferric oxide, sulphur dioxide and sulphur trioxide.
FeSO₄·7H₂O ⟶ FeSO₄ + 7H₂O
2FeSO₄ ⟶ Fe₂O₃ + SO₂ + SO₃
22. (a)
Acids generate hydrogen ions (H⁺) in aqueous solutions, while bases generate hydroxide ions (OH⁻) in aqueous solutions.
(b)
Dry HCl gas does not change the colour of dry litmus paper because hydrogen ions are produced only in the presence of water. In the absence of water, HCl does not show acidic nature.
23.
The main function of veins in the human circulatory system is to carry blood from different parts of the body back to the heart. Veins do not need thick walls because blood flows through them at low pressure.
24. (a)
Proteins control the characteristics of an organism by regulating the chemical processes in the body. For example, in pea plants, the tallness trait is controlled by proteins that help in the synthesis of growth hormones. If the protein is not formed properly, the plant remains dwarf.
(b)
The section of DNA that controls the characteristics of an organism is called a gene.
25. (a)
The student is suffering from myopia (short-sightedness). This defect occurs due to elongation of the eyeball or excessive curvature of the eye lens. It is corrected by using a concave lens.
OR
25. (b)
When a ray of light falls obliquely on a refracting face of a triangular glass prism, it bends towards the normal on entering the prism and away from the normal on emerging out. The angle between the incident ray and the emergent ray is called the angle of deviation.
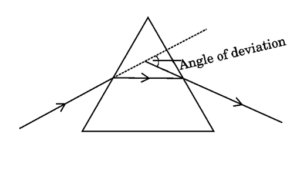
26.
Given,
Voltage (V) = 230 V
Power (P) = 1000 W
Using the relation ![]()
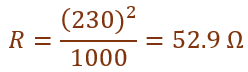
Therefore, the resistance of the heating element is 52.9 ohm.
SECTION C (3 x 7 = 21)
Questions no. 27 to 33 are Short Answer Type questions.
27.
(a) What is a reactivity series of elements ? How is it developed ? Arrange the following elements as they are arranged in the reactivity series :
Aluminium, Calcium, Copper, Lead
(b) Write balanced chemical equation to show the reaction of iron (III) oxide (Fe₂O₃) with aluminium.
28.
(a) Common salt is an important raw material for various chemicals of daily use. State in brief the method of preparation of (i) Sodium hydroxide, and (ii) Sodium hydrogen carbonate from common salt. Write balanced chemical equations of the reactions that occur.
OR
(b) Design an experimental set-up to demonstrate that ‘‘Alcohol and glucose contain hydrogen but are not categorised as acids’’. Also give the reason to justify this fact.
29.
On the basis of the characteristics of the processes given in the brackets in each case, differentiate between the following :
(a) Products of breakdown of pyruvate in aerobic and anaerobic respiration in human beings (product(s) of the processes)
(b) Respiration and photosynthesis in plants (gas released)
(c) Respiration in terrestrial animals and fishes (organs involved)
30.
A pure pea plant having round (R), yellow (Y) seeds is crossed with another pure pea plant having wrinkled (r), green (y) seeds. Subsequently F₁ progeny is self-pollinated to obtain F₂ progeny.
(a) What do the seeds of F₁ generation look like ?
(b) Give the possible combinations of traits in seeds of F₂ generation. Also give their ratio.
(c) State the reason of obtaining seeds of new combination of traits in F₂ generation.
31.
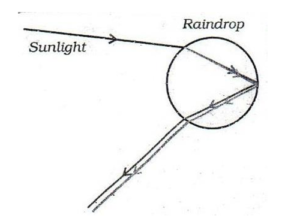
What is a rainbow ? Draw a labelled diagram to show its formation.
32.
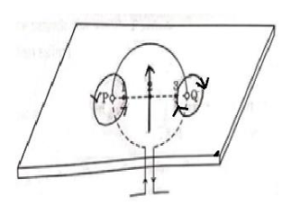
Consider a rectangular cardboard having two holes P and Q through which a current carrying circular loop has been inserted as shown in the diagram.
(a) Make this diagram on your answer sheet and draw three magnetic field lines, one each passing through the points 1 (near P), 2 (at the centre of the loop) and 3 (near Q).
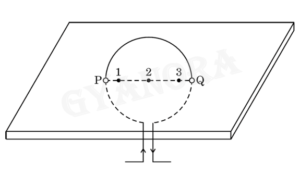
(b) List two factors on which the intensity of the magnetic field produced at the centre of the loop depends.
(c) Name the rule you will apply to determine the direction of magnetic field produced due to a current carrying straight conductor.
33.
(a) ‘‘In a food chain energy flow is unidirectional.’’ Give two reasons for the given statement.
(b) If 10,000 J energy is available at the producer level, how much energy will be available to the secondary consumers ? Give reason to justify your answer.
ANSWERS OF SECTION C
27. (a)
Reactivity series is a list of metals arranged in decreasing order of their chemical reactivity. It is developed by studying the reactions of metals with water, acids and oxygen and by displacement reactions.
According to the reactivity series, the correct order is:
Calcium > Aluminium > Lead > Copper
(b)
When iron (III) oxide reacts with aluminium, aluminium being more reactive displaces iron from its oxide. This reaction is known as the thermite reaction.
Fe₂O₃ + 2Al ⟶ Al₂O₃ + 2Fe
28. (a)
Common salt is used as a raw material for preparing many useful chemicals. Sodium hydroxide is prepared from common salt by the process of electrolysis of brine, known as the chlor-alkali process. During this process, sodium hydroxide, chlorine gas and hydrogen gas are produced.
2NaCl + 2H₂O ⟶ 2NaOH + Cl₂ + H₂
Sodium hydrogen carbonate is prepared from common salt by the Solvay process. Ammonia and carbon dioxide are passed through a concentrated solution of sodium chloride, forming sodium hydrogen carbonate.
NaCl + NH₃ + H₂O + CO₂ ⟶ NaHCO₃ + NH₄Cl
OR
28. (b)
To show that alcohol and glucose contain hydrogen but are not acids, their aqueous solutions are taken in separate test tubes and a metal like zinc is added to each. It is observed that no hydrogen gas is evolved in either case.
This shows that although alcohol and glucose contain hydrogen, they do not produce hydrogen ions in aqueous solution and hence are not categorised as acids.
29.
In aerobic respiration in human beings, pyruvate breaks down completely to form carbon dioxide, water and energy, whereas in anaerobic respiration, pyruvate breaks down to form lactic acid along with a small amount of energy.
In plants, respiration releases carbon dioxide while photosynthesis releases oxygen.
Respiration in terrestrial animals takes place through lungs, whereas in fishes it occurs through gills.
30. (a)
The seeds of the F₁ generation are round and yellow as these traits are dominant over wrinkled and green traits.
(b)
The possible combinations of traits in the F₂ generation are round yellow, round green, wrinkled yellow and wrinkled green seeds in the ratio 9 : 3 : 3 : 1.
(c)
New combinations of traits appear in the F₂ generation due to the independent assortment of genes during gamete formation.
31.
A rainbow is a natural spectrum of sunlight appearing in the sky after rain. It is formed due to the combined effect of refraction, dispersion and total internal reflection of sunlight in water droplets present in the atmosphere.
32. (a)
When a current flows through a circular loop, magnetic field lines are formed around it. These magnetic field lines are circular near the wire and nearly parallel at the centre of the loop.
(b)
The intensity of the magnetic field at the centre of the loop depends on the magnitude of current flowing through the loop and the number of turns of the loop.
(c)
The direction of the magnetic field produced due to a current carrying straight conductor is determined by the Right-hand thumb rule.
33. (a)
In a food chain, energy flow is unidirectional because energy is transferred from producers to consumers and is not recycled back. Also, a large amount of energy is lost as heat at each trophic level.
(b)
According to the 10% law of energy transfer, only 10% of energy is passed to the next trophic level. Therefore, from 10,000 J at the producer level, 100 J of energy will be available to the secondary consumers.
SECTION D (3 x 5 = 15)
Questions no. 34 to 36 are Long Answer Type questions.
34.
(a) (i) Consider the following metals :
K, Ca, Al, Cu, Ag, Fe
Select from the above metals, a metal which
- does not react with oxygen even at high temperature.
- reacts with oxygen at ordinary temperature and forms a protective oxide layer which prevents the metal from further oxidation.
III. catches fire when kept in the open.
- does not burn in oxygen but the hot metal is coated with a black coloured oxide layer.
(ii) What are amphoteric oxides ? With the help of balanced chemical equations show that aluminium oxide is an amphoteric oxide.
(iii) What are alkalis ? Give one example.
OR
(b) (i) With the help of balanced chemical equations state the process of extracting
(I) mercury from its ore called cinnabar, and
(II) copper from its sulphide ore.
(ii) Silver and copper articles slowly lose their shiny surfaceswhen exposed to air. Name the compounds formed on (I) silver articles, and (II) copper articles in the form of coating.
35.
(a) (i) Identify the parts ‘X’ and ‘Y’ in the figure given below:
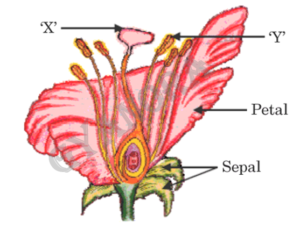
(ii) Name the yellowish coloured structures produced by the part labelled as ‘Y’.
(iii) Write the name of the process by which these are transferred to the part labelled as ‘X’.
(iv) Explain the process of seed formation in a flowering plant.
OR
(b) (i) Name the type of asexual mode of reproduction shown in the given figure.
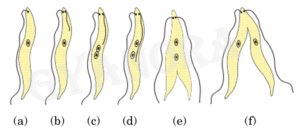
(ii) Identify the unicellular organism in the diagram.
(iii) List any two advantages of asexual reproduction over sexual reproduction.
(iv) Name and explain any one mode of asexual reproduction observed in Hydra.
36.
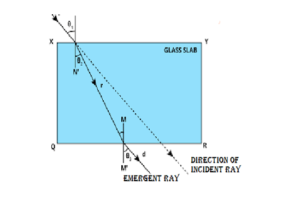
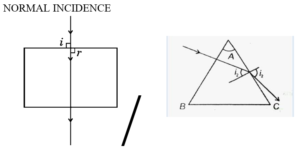
(a) (i) ‘‘In refraction of light through a rectangular glass slab, the emergent ray is always parallel to the direction of the incident ray.’’ Why ? Explain with the help of a ray diagram. What happens when a ray of light falls normally on one of the faces of a rectangular glass prism ? Draw diagram.
(ii) An object is placed at a distance of 30 cm from the optical centre of a concave lens of focal length 20 cm. Use Lens formula to determine the position of the image formed in this case.
OR
(b) (i) A student wishes to study the image formation by a concave mirror using candle flame as object. State the type of the image formed by the mirror and mention the change in the image formed, if any, that he observes when the candle flame is gradually moved away from the pole of the mirror. Draw a ray diagram to show the image formation when the object distance is nearly equal to the radius of curvature of the mirror.
(ii) A convex mirror used for rear-view on an automobile has a focal length of 3.0 m. If a bus is located at 6.0 m from this mirror, use mirror formula to find the position of the image of the bus as seen in the mirror.
ANSWERS OF SECTION-D
34. (a)
(i)
I. Silver (Ag) does not react with oxygen even at high temperature.
II. Aluminium (Al) reacts with oxygen at ordinary temperature and forms a thin protective layer of aluminium oxide which prevents further oxidation.
III. Potassium (K) catches fire when kept in the open due to its high reactivity with oxygen and moisture.
IV. Iron (Fe) does not burn in oxygen, but when heated strongly, it gets coated with a black coloured oxide layer of iron oxide.
(ii)
Amphoteric oxides are oxides which react with both acids and bases to form salt and water. Aluminium oxide is an amphoteric oxide because it reacts with acids as well as bases.
With acid:
Al₂O₃ + 6HCl ⟶ 2AlCl₃ + 3H₂O
With base:
Al₂O₃ + 2NaOH ⟶ 2NaAlO₂ + H₂O
(iii)
Alkalis are bases which are soluble in water and produce hydroxide ions in aqueous solutions. One example of an alkali is sodium hydroxide (NaOH).
OR
34. (b)
(i)
Mercury is extracted from its sulphide ore cinnabar by roasting it in air. During roasting, cinnabar decomposes to form mercury and sulphur dioxide.
2HgS + 3O₂ ⟶ 2HgO + 2SO₂
On further heating, mercury oxide decomposes to give mercury.
2HgO ⟶ 2Hg + O₂
Copper is extracted from its sulphide ore by heating it strongly in air. Copper sulphide is first converted into copper oxide, which then reacts with copper sulphide to produce copper metal.
2Cu₂S + 3O₂ ⟶ 2Cu₂O + 2SO₂
Cu₂S + 2Cu₂O ⟶ 6Cu + SO₂
(ii)
Silver articles get coated with silver sulphide (Ag₂S) due to reaction with sulphur compounds present in air. Copper articles get coated with a green layer of basic copper carbonate when exposed to moist air.
35. (a)
(i)
In the given figure, part X is the ovary and part Y is the anther.
(ii)
The yellowish coloured structures produced by the anther are called pollen grains.
(iii)
The process by which pollen grains are transferred from anther to stigma is called pollination.
(iv)
After pollination, fertilisation takes place inside the ovary where the male gamete fuses with the female gamete to form a zygote. The zygote develops into an embryo, while the ovule forms the seed and the ovary develops into a fruit. This entire process leads to seed formation in flowering plants.
OR
35. (b)
(i)
The type of asexual reproduction shown in the given figure is binary fission.
(ii)
The unicellular organism shown in the diagram is Amoeba.
(iii)
Two advantages of asexual reproduction are that it requires only one parent and it results in rapid multiplication of organisms.
(iv)
In Hydra, asexual reproduction occurs by budding. In this process, a small outgrowth develops on the body of the parent, grows into a new individual and finally detaches to live independently.
36. (a)
(i)
In refraction through a rectangular glass slab, the emergent ray is always parallel to the incident ray because the angle of refraction at the first surface is equal to the angle of emergence at the second surface, as the opposite faces of the slab are parallel. When a ray of light falls normally on one face of a rectangular glass slab, it passes straight through without any deviation.
(ii)
Given,
Object distance (u) = –30 cm
Focal length (f) = –20 cm
Using lens formula,
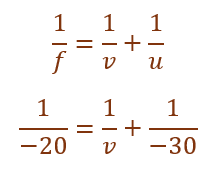
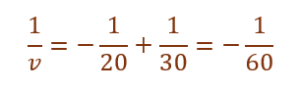
v = –60 cm
Thus, the image is formed 60 cm in front of the lens.
OR
36. (b)
(i)
A concave mirror forms a real and inverted image when the object is placed beyond the focal point. As the candle flame is moved away from the pole of the mirror, the image gradually shifts towards the focus and becomes smaller in size. When the object is placed nearly at the radius of curvature, the image is formed at the same distance, real, inverted and of the same size.
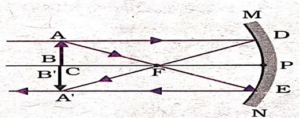
(ii)
Given,
Focal length (f) = +3.0 m
Object distance (u) = –6.0 m
Using mirror formula,
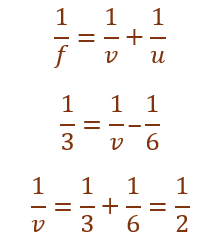
v = +2 m
Thus, the image is formed 2 m behind the mirror.
SECTION E (3 x 4 = 12)
The following questions are Source-based/Case-based questions. Read the case carefully and answer the questions that follow.
37.
‘A’ and ‘B’ are two salts used for washing purposes. Salt ‘A’ is used for bathing also. Four test tubes I, II, III and IV as mentioned below are taken.
- Rain water + solution of salt ‘A’
- Rain water + solution of salt ‘B’
III. Tubewell water + solution of salt ‘A’
- Tubewell water + solution of salt ‘B’
The test tubes are shaken one by one almost identically for the same time and the lengths of foam formed in each test tube is noted.
(a) In which one of the four test tubes is the foam formed the minimum ? (1)
(b) Differentiate between salt A and salt B. (1)
(c) (i) What are esters ? What happens when an ester reacts with an alkali (say sodium hydroxide) ? Give chemical equation for the reaction. (2)
OR
(c) (ii) What is the cause of hardness of water ? Sometimes it is observed that while bathing foam is formed with difficulty and an insoluble substance is formed. Name this substance and write the cause of its formation. (2)
38.
A person while climbing up a rocky hill comes into a panic state and fear. His body starts reacting in a ‘‘flight-or-flight’’ condition to adjust to the dangerous and stressful situation. Based on the above facts, answer the questions that follow.
(a) (i) Name the hormone secreted in the blood of the person in this situation. (2)
OR
(a) (ii) Name the source gland of the hormone secreted in this condition. (2)
(b) State any two responses in the body of the person as a result of the secretion of this hormone. (1)
(c) How does the action of the chemical signal in terms of hormones differ from the electrical impulses via nerve cells ? (1)
39.
As shown in the diagram, an electric circuit consisting of an ammeter, a voltmeter, 4 cells of 1.5 V each, a plug key with a gap XY was set up. Voltmeter and ammeter readings were recorded in the observation table for four arrangements as given below :
Arrangement No. 1 – only resistor R₁ in gap XY
Arrangement No. 2 – only resistor R₂ in gap XY
Arrangement No. 3 – Resistors R₁ and R₂ in series in gap XY
Arrangement No. 4 – Resistors R₁ and R₂ in parallel in gap XY
Based on the observations, four V–I graphs A, B, C and D as shown in figure were drawn. Study these graphs.
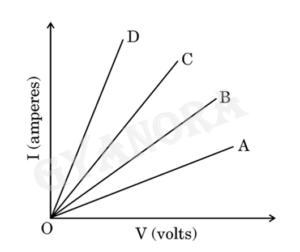
(a) Which one of the graphs represents the series combination of R₁ and R₂ ? (1)
(b) Which one of these graphs represents the parallel combination of R₁ and R₂ ? (1)
(c) (i) Show an arrangement of three resistors, each of resistance 10 Ω, so that the combination has a resistance of 15 Ω. Give justification for your answer. (2)
OR
(c) (ii) A battery of 6 V is connected with a series combination of five resistors of 0.1 Ω, 0.2 Ω, 0.3 Ω, 0.4 Ω and 0.5 Ω. How much current would flow through the 0.3 Ω resistor ? Justify your answer. (2)
ANSWERS OF SECTION-E
37.
(a)
Minimum foam is formed in test tube IV (Tubewell water + solution of salt ‘B’) because tubewell water is hard water and salt ‘B’ does not form lather easily in hard water.
(b)
Salt A is a soap which forms lather easily in soft water and can be used for bathing, whereas salt B is a detergent which forms lather even in hard water and is mainly used for washing clothes.
(c) (i)
Esters are sweet-smelling organic compounds formed by the reaction of an acid with an alcohol. When an ester reacts with an alkali like sodium hydroxide, it undergoes saponification to form an alcohol and a salt of the acid.
CH₃COOC₂H₅ + NaOH ⟶ CH₃COONa + C₂H₅OH
OR
(c) (ii)
Hardness of water is caused due to the presence of calcium and magnesium salts dissolved in it. While bathing with hard water, soap reacts with these salts to form an insoluble substance called scum, which prevents the formation of lather.
38.
(a) (i)
The hormone secreted into the blood in this situation is adrenaline.
OR
(a) (ii)
The hormone is secreted by the adrenal gland.
(b)
Due to the secretion of adrenaline, the heart rate increases and more blood is supplied to the muscles, preparing the body to face the emergency situation.
(c)
Hormones act slowly and their effect lasts for a longer duration as they are transported through blood, whereas electrical impulses act very fast through nerve cells and their effect lasts for a very short time.
39.
(a)
The graph which represents the series combination of R₁ and R₂ is the one showing least current for a given voltage, because resistance is maximum in series combination.
(b)
The graph which represents the parallel combination of R₁ and R₂ is the one showing maximum current for a given voltage, because resistance is minimum in parallel combination.
(c) (i)
To obtain a resistance of 15 Ω using three resistors of 10 Ω each, two resistors are connected in parallel to give an equivalent resistance of 5 Ω, and this combination is connected in series with the third 10 Ω resistor. Thus, total resistance becomes 15 Ω.
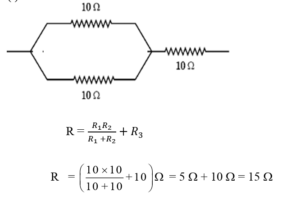
OR
(c) (ii)
In a series combination, the same current flows through all resistors. The total resistance of the circuit is
0.1 + 0.2 + 0.3 + 0.4 + 0.5 = 1.5 Ω.
Using Ohm’s law,
I = V/R = 6/1.5 = 4 A.
Therefore, a current of 4 ampere flows through the 0.3 Ω resistor.
x——————x—————–x——————x
Revise your Science Concepts for Board Exam by CLICK HERE or visit Gyanora.in
Practice Chapter-Wise Extra Questions of Science for Your Board Exams by CLICK HERE
OR Explore other Chapter’s Extra Questions:
Chapter-2 Acid, Bases, and Salt
Chapter-3 Metals and Non-Metals
Chapter-4 Carbon and it’s Compounds
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6



