PYQ 2024 SET-1
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections – A, B, C, D and E.
- Section A – Questions No. 1 to 20 are Multiple Choice Questions. Each question carries 1 mark.
- Section B – Questions No. 21 to 26 are Very Short Answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C – Questions No. 27 to 33 are Short Answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D – Questions No. 34 to 36 are Long Answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E – Questions No. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
SECTION – A
Select and write the most appropriate option out of the four given options for each of the questions 1–20. There is no negative mark for incorrect response.
1. When 2 mL of sodium hydroxide solution is added to few pieces of aluminium, it is found that the reaction cannot take place even on heating. Which of the following chemical equation is correct?
(a) NaOH + Zn → Na₂ZnO₂ + H₂O
(b) 2NaOH + Zn → Na₂ZnO₂ + H₂
(c) 2NaOH + Zn → Na₂ZnO₂ + H₂O
(d) 2NaOH + Zn → Na₂ZnO₂ + H₂ + H₂O
2. Select from the following a decomposition reaction in which source of energy for decomposition is light:
(a) FeSO₄ → Fe₂O₃ + SO₂ + SO₃
(b) 2H₂O → 2H₂ + O₂
(c) 2AgCl → 2Ag + Cl₂
(d) CaCO₃ → CaO + CO₂
3. A metal and a non-metal that exists in liquid state at the room temperature are respectively:
(a) Bromine and Mercury
(b) Mercury and Iodine
(c) Mercury and Bromine
(d) Iodine and Mercury
4. Carbon compounds:
(i) are good conductors of electricity.
(ii) are bad conductors of electricity.
(iii) have strong forces of attraction between their molecules.
(iv) have weak forces of attraction between their molecules.
The correct statements are:
(a) (i) and (iii)
(b) (ii) and (iv)
(c) (i) and (iv)
(d) (ii) and (iii)
5. Consider the following compounds:
FeSO₄, CuSO₄, Na₂CO₃
The compound forming maximum number of water of crystallisation in its crystalline form is:
(a) FeSO₄
(b) CuSO₄
(c) Na₂CO₃
(d) CaSO₄
6. Oxides of aluminium and zinc are:
(a) acidic
(b) basic
(c) amphoteric
(d) neutral
7. MnO₂ + 4HCl → MnCl₂ + 2H₂O + Cl₂
The reaction given above is a redox reaction because in this case:
(a) MnO₂ is oxidised and HCl is reduced.
(b) MnO₂ is oxidised.
(c) MnO₂ is reduced and HCl is oxidised.
(d) MnO₂ is reduced.
8. Consider the following statements:
(i) The sex of a child is determined by what it inherits from the mother.
(ii) The sex of a child is determined by what it inherits from the father.
(iii) The probability of having a male or a female child is same.
(iv) The sex of a child is determined at the time of fertilisation when male and female gametes fuse to form a zygote.
The correct statements are:
(a) (i) and (ii)
(b) (ii) and (iv)
(c) (iii) and (iv)
(d) (ii), (iii) and (iv)
9. Chromosomes:
(i) carry hereditary information from parents to the next generation.
(ii) are thread-like structures located inside the nucleus of an animal cell.
(iii) always exist in pairs in human reproductive cells.
(iv) are associated with the process of cell division.
The correct statements are:
(a) (i) and (ii)
(b) (ii) and (iv)
(c) (i), (ii) and (iv)
(d) (i) and (iv)
10. In a nerve cell, the site where the electrical impulse is converted into a chemical signal is known as:
(a) Axon
(b) Dendrites
(c) Neuromuscular junction
(d) Cell body
11. A stoma closes when:
(i) it needs carbon dioxide for photosynthesis.
(ii) it does not need carbon dioxide for photosynthesis.
(iii) water flows out of the guard cells.
(iv) water flows into the guard cells.
The correct reasons in this process are:
(a) (i) only
(b) (i) and (iii)
(c) (ii) and (iii)
(d) (ii) and (iv)
12. At what distance from a convex lens should an object be placed to get an image of the same size but of opposite direction as screen?
(a) Beyond twice the focal length of the lens
(b) At the principal focus of the lens
(c) At twice the focal length of the lens
(d) Between the optical centre of the lens and its principal focus
13. The lens system by which an eye forms an image on a light sensitive screen, which is called as:
(a) Cornea
(b) Ciliary muscles
(c) Optic nerve
(d) Retina
14. The pattern of the magnetic field produced inside a current carrying solenoid is:
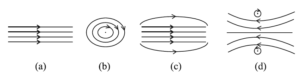
15. Identify the food chain in which the organisms of the second trophic level are missing:
(a) Grass, goat, lion
(b) Zooplankton, phytoplankton, small fish, large fish
(c) Tiger, grass, snake, frog, eagle
(d) Grasshopper, grass, snake, frog, eagle
16. In which of the following organisms, multiple fission is a means of asexual reproduction?
(a) Yeast
(b) Leishmania
(c) Planaria
(d) Plasmodium
For Q. no. 17 to 20, two statements are given – one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below:
(a) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(b) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
(c) Assertion (A) is true, but Reason (R) is false.
(d) Assertion (A) is false, but Reason (R) is true.
17. Assertion (A): Hydrogen gas is not evolved when zinc reacts with nitric acid.
Reason (R): Nitric acid oxidises the hydrogen gas produced to water.
18. Assertion (A): Accumulation of harmful chemicals increases in the organisms at the higher trophic level of a food chain.
Reason (R): Harmful chemicals are sprayed on the crops to protect them from diseases and pests.
19. Assertion (A): The rate of breathing in aquatic organisms is much faster than in terrestrial organisms.
Reason (R): The amount of oxygen dissolved in water is very high as compared to the amount of oxygen in air.
20. Assertion (A): The rainbow is a natural spectrum of sunlight in the sky.
Reason (R): Rainbow is formed in the sky when the sun is overhead and water droplets are also present in air.
ANSWERS OF SECTION – A
- (d)
2. (c)
3. (c)
4. (c)
5. (a)
6. (c)
7. (d)
8. (c)
9. (c)
10. (c)
11. (c)
12. (c)
13. (d)
14. (a)
15. (c)
16. (d)
17. (a)
18. (a)
19. (c)
20. (c)
SECTION – B (2 x 6 = 12)
Questions no. 21 to 26 are Very Short Answer Type questions.
21. Name the type of chemical reaction in which calcium oxide reacts with water. Justify your answer by giving balanced chemical equation for the chemical reaction.
22. State one role of each of the following in human digestive system:
(i) Hydrochloric acid
(ii) Villi
(iii) Salivary amylase
(iv) Lipase
23. (A) How is the movement of leaves of sensitive plant different from the downward movement of roots?
OR
(B) There is a hormone which regulates carbohydrate, protein and fat metabolism in our body. Name the hormone and the gland which secretes it. Why is the deficiency of this hormone harmful in our body?
24. An object is placed at a distance of 10 cm from a convex mirror of focal length 15 cm. Find the position of the image formed by the mirror.
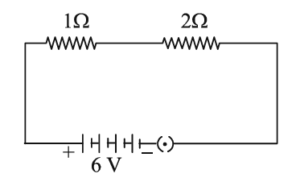
25. (A) How many two coiled cement-free resistors out of resistance 6 Ω, so that the combination has a resistance of 9 Ω? Also justify your answer.
OR
(B) In the given circuit calculate the power consumed in watts in the resistor of 2 Ω.
26. (i) Two magnetic field lines do not intersect each other. Why?
(ii) How is a uniform magnetic field in a given region represented? Draw a diagram in support of your answer.
ANSWERS OF SECTION – B
21.
Calcium oxide reacts with water to form calcium hydroxide. This reaction is a combination reaction because two substances combine to form a single product.
Balanced chemical equation:
CaO + H₂O → Ca(OH)₂
22.
(i) Hydrochloric acid: It creates an acidic medium in the stomach which helps in digestion of proteins and kills harmful bacteria.
(ii) Villi: They increase the surface area of the small intestine for absorption of digested food.
(iii) Salivary amylase: It digests starch into simpler sugars.
(iv) Lipase: It helps in digestion of fats into fatty acids and glycerol.
23. (A)
The movement of leaves of sensitive plant is a nastic movement, which is non-directional and occurs due to change in turgor pressure.
The downward movement of roots is a tropic movement, which is directional and occurs due to growth.
OR
23. (B)
The hormone is Insulin, which is secreted by the pancreas.
Deficiency of insulin causes increase in blood sugar level, leading to diabetes, which is harmful for the body.
24.
Given:
Focal length of convex mirror, f = +15 cm
Object distance, u = –10 cm
Using mirror formula:
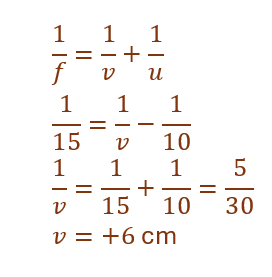
Thus, the image is formed 6 cm behind the mirror.
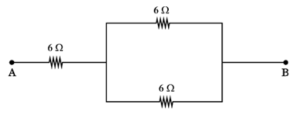
25. (A)
To obtain a resistance of 9 Ω using 6 Ω resistors, two resistors are connected in parallel and then in series with another 6 Ω resistor.
This combination gives the required resistance of 9 Ω.
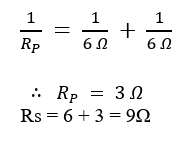
OR
25. (B)
Using the given circuit, the power consumed in the 2 Ω resistor is calculated using:

26.
(i) Two magnetic field lines do not intersect because at the point of intersection the magnetic field would have two directions, which is not possible.
(ii) A uniform magnetic field is represented by parallel and equally spaced straight lines, all pointing in the same direction.

SECTION – C (3 x 7 = 21)
Questions no. 27 to 33 are Short Answer Type questions.
27Write one chemical equation each for chemical reaction in which the following have taken place:
(i) Change in colour
(ii) Change in temperature
(iii) Formation of precipitate
Mention colour change/temperature change/gas/solid compound produced in each with equation.
28. (i) The pH of a sample of pure juice is 4.6. How is this juice likely to taste? Give reason to justify your answer.
(ii) How do we differentiate between a strong acid and a weak acid in terms of ionisation in aqueous solutions?
(iii) The acid rain can make the survival of aquatic animals difficult. How?
29. (i) Why is respiratory pigment needed in multicellular organisms with large body size?
(ii) Give reasons for the following:
(a) Rings of cartilage are present in the trachea.
(b) Lungs always contain a residual volume of air.
(c) The diaphragm flattens and ribs are lifted up when we breathe in.
(iii) Walls of alveoli contain an extensive network of blood vessels.
30. Define reflex action. With the help of a flow chart show the path of a reflex action such as sneezing.
31. Study the diagram given below and answer the questions that follow:
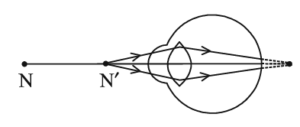
(i) Name the defect of vision represented in the diagram. Give reason for your answer.
(ii) List two causes of this defect.
(iii) With the help of a diagram show how this defect of vision is corrected.
32. Name and state the rule to determine the direction of a :
(i) magnetic field produced around a current carrying straight conductor.
(ii) force experienced by a current carrying straight conductor placed in a magnetic field which is perpendicular to it.
33. (A) Plants → Deer → Lion
In the given food chain, what will be the impact of removing all the organisms of second trophic level on the first and third trophic level ?
Will the impact be same for the organisms of third trophic level in the above food chain if they were present in a food web ?
Justify.
OR
(B) A gas ‘X’ which is a deadly poison is found at the higher levels of atmosphere and performs an essential function.
Name the gas and write the essential function performed by this gas in the atmosphere. Which chemical is linked to the decrease in the level of this gas ? What measures have been taken by an international organization to check the depletion of the layer containing this gas ?
ANSWERS OF SECTION – C
27.
(i) Change in colour:
When an iron nail is placed in copper sulphate solution, iron displaces copper from the solution. As a result, the blue colour of copper sulphate changes to green due to formation of iron sulphate.
Fe + CuSO₄ → FeSO₄ + Cu
(ii) Change in temperature:
When calcium oxide reacts with water, calcium hydroxide is formed and heat is released. Therefore, the temperature of the reaction mixture increases.
CaO + H₂O → Ca(OH)₂ + Heat
(iii) Formation of precipitate:
When sodium sulphate reacts with barium chloride, a white insoluble precipitate of barium sulphate is formed.
Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
28.
(i) The tomato juice will taste sour because its pH value is 4.6, which is less than 7, showing that it is acidic in nature.
(ii) A strong acid produces a large number of ions in aqueous solution, whereas a weak base produces comparatively fewer ions in aqueous solution.
(iii) Acid rain lowers the pH of water in lakes and rivers, which can damage the body functions of aquatic animals and may even cause their death.
29.
(i) Respiratory pigment is needed in multicellular organisms with large body size because simple diffusion is not sufficient to transport oxygen to all body cells.
(ii) Rings of cartilage are present in the throat to prevent the trachea from collapsing. Lungs always contain a residual volume of air to allow continuous exchange of gases. When we breathe in, the diaphragm flattens and ribs are lifted up, increasing the volume of the chest cavity. Walls of alveoli contain an extensive network of blood vessels to allow rapid exchange of oxygen and carbon dioxide.
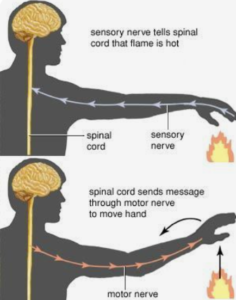
30.
A reflex action is a quick, automatic and involuntary response of the body to a stimulus.
The path of a reflex action is: stimulus is received by receptors, which send impulses through sensory neurons to the spinal cord. From the spinal cord, impulses are transmitted through motor neurons to the effector organ, producing a response such as sneezing.
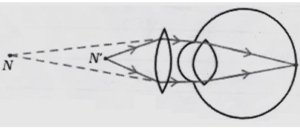
31.
(i) The defect of vision shown in the diagram is myopia (short-sightedness) because the image of a distant object is formed in front of the retina.
(ii) The two main causes of myopia are elongation of the eyeball and excessive curvature of the eye lens.
(iii) This defect is corrected by using a concave lens, which diverges the light rays and helps the image to form on the retina.
32.
(i) The direction of magnetic field around a current-carrying straight conductor is determined by the right-hand thumb rule. According to this rule, if the thumb of the right hand points in the direction of current, the curled fingers show the direction of the magnetic field.
(ii) The direction of force on a current-carrying conductor placed in a magnetic field is determined by Fleming’s left-hand rule. According to this rule, the thumb, forefinger and middle finger of the left hand are held mutually perpendicular to each other, where forefinger represents magnetic field, middle finger represents current and thumb shows the direction of force.
33. (A)
If all the organisms of the second trophic level are removed, the population of plants will increase due to absence of grazing, while the population of lions will decrease due to lack of food.
If the organisms of the third trophic level were part of a food web, the impact would be less severe because they would have alternative food sources available.
OR
33. (B)
The gas ‘X’ is ozone. It performs the essential function of absorbing harmful ultraviolet radiation from the sun and protects living organisms on Earth. Chlorofluorocarbons (CFCs) are responsible for the depletion of the ozone layer. International agreements such as the Montreal Protocol have been adopted to control the use of CFCs and protect the ozone layer.
SECTION – D (3 x 5 = 15)
Questions no. 34 to 36 are Long Answer Type questions.
34. (A)
(i) Define a homologous series of carbon compounds.
(ii) Why is the melting and boiling points of C₄H₁₀ higher than that of C₂H₆ or C₄H₈ ?
(iii) Why do we NOT see any gradation in chemical properties of a homologous series compounds ?
(iv) Write the name and structures of (i) aldehyde and (ii) ketone with molecular form C₃H₆O.
OR
(B)
(i) Write the name and structure of an organic compound ‘X’ having two carbon atoms in its molecule and its name is suffixed with ‘–ol’.
(ii) What happens when ‘X’ is heated with excess concentrated sulphuric acid at 443 K ? Write chemical equation for the reaction stating the conditions for the reaction. Also state the role played by concentrated sulphuric acid in the reaction.
(iii) Name and draw the electron dot structure of hydrocarbon produced in the above reaction.
35. (A)
(i) Name three techniques/devices used by human females to avoid pregnancy. Mention the side effects caused by each.
(ii) What will happen if in a human female (a) fertilisation takes place, (b) an egg is not fertilised ?
OR
(B)
(i) Draw a diagram showing spore formation in Rhizopus and label the (a) reproductive and (b) non-reproductive parts. Why does Rhizopus not multiply on a dry slice of bread ?
(ii) Name and explain the process by which reproduction takes place in Hydra.
36. (A)
(i) Define electric power. Express it in terms of potential difference (V) and resistance (R). 5
(ii) An electric oven is designed to work on the mains voltage of 220 V. This oven consumes 11 units of electrical energy in 5 hours. Calculate :
(a) power rating of the oven
(b) current drawn by the oven
(c) resistance of the oven when it is red hot
OR
(B)
(i) Write the relation between resistance R and electrical resistivity ρ of the material of a conductor in the shape of cylinder of length l and area of cross-section A. Hence derive the SI unit of electrical resistivity.
(ii) The resistance of a metal wire of length 3 m is 60 Ω. If the area of cross-section of the wire is 4 × 10⁻⁷ m², calculate the electrical resistivity of the wire.
(iii) State how would electrical resistivity be affected if the wire (of part (ii)) is stretched so that its length is doubled. Justify your answer.
ANSWERS OF SECTION – D
34. (A)
A homologous series is a group of organic compounds having the same general formula, similar chemical properties and a gradual change in physical properties.
The melting and boiling points of C₄H₁₀ are higher than those of smaller hydrocarbons because it has a higher molecular mass and stronger intermolecular forces.
No gradation is seen in chemical properties of a homologous series because all members contain the same functional group and undergo similar chemical reactions.
An aldehyde with molecular formula C₃H₆O is propanal (CH₃CH₂CHO).
A ketone with molecular formula C₃H₆O is propanone (CH₃COCH₃).
OR
34. (B)
The organic compound ‘X’ is ethanol (C₂H₅OH). Its structure contains two carbon atoms and the suffix ‘–ol’ indicates alcohol.
When ethanol is heated with excess concentrated sulphuric acid at 443 K, it undergoes dehydration to form ethene.
C₂H₅OH → C₂H₄ + H₂O
Concentrated sulphuric acid acts as a dehydrating agent in this reaction.
The hydrocarbon formed is ethene (C₂H₄). Its electron dot structure shows a double bond between the two carbon atoms.
35. (A)
Three methods used by human females to avoid pregnancy are oral contraceptive pills, intrauterine devices and surgical methods. Oral pills may cause hormonal imbalance, IUCDs may cause irritation, and surgical methods are irreversible.
If fertilisation takes place, a zygote is formed which develops into an embryo. If the egg is not fertilised, the uterine lining breaks down and menstruation occurs.
OR
35. (B)
(i) Rhizopus reproduces by spore formation. Spores are produced inside sporangia, which is the reproductive part, while hyphae form the non-reproductive part. Rhizopus does not multiply on dry bread because moisture is essential for spore germination.
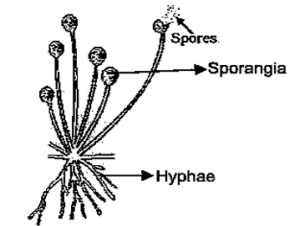
(ii) Hydra reproduces by budding, in which a small outgrowth develops on the parent body and later detaches to form a new individual.
36. (A)
Electric power is the rate at which electrical energy is consumed in a circuit.
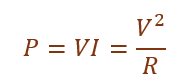
Given:
Energy = 11 units, Time = 5 h
Power = 11/5 = 2.2 kW
Current = P/V = 2200/220 = 10 A
Resistance = V/I = 22 Ω
OR
36. (B)
The relation between resistance and resistivity is:
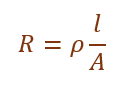
The SI unit of resistivity is ohm metre (Ω m).
Given:
R = 60 Ω, l = 3 m, A = 4 × 10⁻⁷ m²
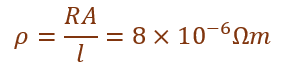
If the wire is stretched to double its length, its resistivity remains unchanged because resistivity depends only on the nature of material and temperature.
SECTION – E (3 x 4 = 12)
Nos. 37–39 are source-based/case-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
37. The metals produced by various reduction processes are not very pure. They contain impurities, which must be removed to obtain pure metals. The most widely used method for refining impure metals is electrolytic refining.
(i) What is the cathode and anode made of in the refining of copper by this process ? (1)
(ii) Name the solution used in the above process and write its formula. (1)
(iii) (A) How copper gets refined when electric current is passed in the electrolytic cell ? (2)
OR
(iii) (B) You have two beakers ‘A’ and ‘B’ containing copper sulphate solution. What would you observe after about 2 hours if you dip a strip of zinc in beaker ‘A’ and a strip of silver in beaker ‘B’ ? Give reason for your observations in each case. (2)
38. Mendel worked out the rules of heredity by working on garden pea using a number of visible contrasting characters. He conducted several experiments by making a cross with one or two pairs of contrasting characters of pea plant. On the basis of his observations he gave some interpretations which helped to study the mechanism of inheritance.
(i) When Mendel crossed pea plants with pure tall and pure short characteristics to produce F₁ progeny, which two observations were made by him in F₁ plants ? (1)
(ii) Write one difference between dominant and recessive trait. (1)
(iii) (A) In a cross with two pairs of contrasting characters
RRY Y × rryy
(Round Yellow) (Wrinkled Green)
Mendel observed 4 types of combinations in F₂ generation. By which method did he obtain F₂ generation ? Write the ratio of the parental combinations obtained and what conclusions were drawn from this experiment. (2)
OR
(iii) (B) Justify the statement :
“It is possible that a trait is inherited but may not be expressed.” (2)
39. Study the data given below showing the focal length of three concave mirrors A, B and C and the respective distances of objects placed in front of the mirrors :
| Case | Mirror | Focal Length (cm) | Object Distance (cm) |
| 1 | A | 20 | 45 |
| 2 | B | 15 | 30 |
| 3 | C | 30 | 20 |
(i) In which one of the above cases the mirror will form a diminished image of the object ? Justify your answer. (1)
(ii) List two properties of the image formed in case 2. (2)
(iii) (A) What is the nature and size of the image formed by mirror C ? Draw ray diagram to justify your answer. (2)
OR
(iii) (B) An object is placed at a distance of 18 cm from the pole of a concave mirror of focal length 12 cm. Find the position of the image formed in this case. (2)
ANSWERS OF SECTION – E
37.
(i) In electrolytic refining of copper, the anode is made of impure copper and the cathode is a thin sheet of pure copper.
(ii) The solution used is acidified copper sulphate solution (CuSO₄).
(iii) (A) When electric current is passed, copper from the anode dissolves into the solution as copper ions. These ions move towards the cathode and get deposited as pure copper, while impurities fall down as anode mud.
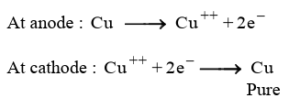
OR
(iii) (B) Zinc displaces copper from copper sulphate solution in beaker A because zinc is more reactive than copper. In beaker B, silver does not displace copper because silver is less reactive than copper, so no change is observed.
38.
(i) Mendel observed that all plants in the F₁ generation were tall and the dwarf character did not appear.
(ii) A dominant trait expresses itself in the presence of its contrasting trait, whereas a recessive trait expresses only in the absence of the dominant trait.
(iii) (A) Mendel obtained the F₂ generation by self-pollination of F₁ plants. The parental combinations appeared in the ratio 9 : 3 : 3 : 1, which led to the conclusion that traits assort independently during gamete formation.
OR
(iii) (B) A trait may be inherited but not expressed if it is recessive and masked by a dominant trait in the organism.
39.
(i) In case 1, the mirror forms a diminished image because the object is placed beyond the centre of curvature of the concave mirror.
(ii) In case 2, the image formed is real and inverted.
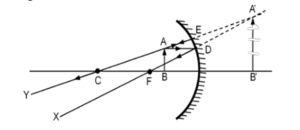
(iii) (A) In mirror C, the image formed is virtual and enlarged because the object is placed between the pole and the focus. A proper ray diagram should be drawn to justify this.
OR
(iii) (B)
Using mirror formula:
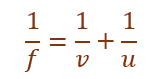
Given:
f = –12 cm, u = –18 cm
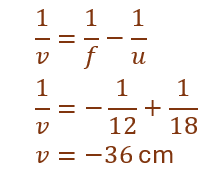
Thus, the image is formed 36 cm in front of the mirror.
x——————x—————–x——————x
Revise your Science Concepts for Board Exam by CLICK HERE or visit Gyanora.in
Practice Chapter-Wise Extra Questions of Science for Your Board Exams by CLICK HERE
OR Explore other Chapter’s Extra Questions:
Chapter-2 Acid, Bases, and Salt
Chapter-3 Metals and Non-Metals
Chapter-4 Carbon and it’s Compounds
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6



