PYQ PAPER 2025
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections – A, B, C, D and E.
- Section A – Questions No. 1 to 20 are Multiple Choice Questions. Each question carries 1 mark.
- Section B – Questions No. 21 to 26 are Very Short Answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C – Questions No. 27 to 33 are Short Answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D – Questions No. 34 to 36 are Long Answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E – Questions No. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
SECTION A (20 × 1 = 20)
Questions No. 1 to 20 are Multiple Choice Questions. Only one of the choices is correct. Select and write the most appropriate option out of the four options given for each.
1. The correct balanced chemical equation showing exothermic reaction in which natural gas burns in air is :
(A) CH₄ + O₂ ⎯→ CO₂ + 2H₂O
(B) CH₄ + 2O₂ ⎯→ 2CO₂ + 2H₂O + Energy
(C) CH₄ + 2O₂ ⎯→ CO₂ + 2H₂O
(D) CH₄ + 2O₂ ⎯→ CO₂ + 2H₂O + Energy
2. The warning sign shown in the given figure must invariably be displayed/pasted on the containers which contain hydroxide of:
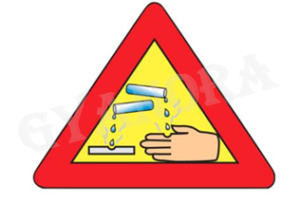
(A) Aluminium
(B) Calcium
(C) Sodium
(D) Magnesium
3. The body of human beings works within the pH range of :
(A) 6.1 to 6.8
(B) 6.5 to 7.3
(C) 7.0 to 7.8
(D) 7.5 to 8.1
4. Aluminium powder is used in thermit welding because :
(A) Its reaction with iron is highly exothermic.
(B) When it is heated with iron (III) oxide, molten iron is obtained.
(C) When it is heated with iron (III) oxide, molten aluminium oxide is obtained to join railway tracks.
(D) Its melting point is low as compared to iron and a molten alloy of iron and aluminium is formed on heating which is used to join railway tracks.
5. Two metals zinc and tin are dissolved separately in definite proportions in molten copper (the primary metal) to obtain two different alloys respectively known as :
(A) Bronze and Brass
(B) Brass and Solder
(C) Brass and Bronze
(D) Solder and Bronze
6. The opening and closing of stomata is regulated by :
(A) CO₂ concentration in stomata
(B) Temperature in guard cells
(C) O₂ concentration in stomata
(D) Amount of water in guard cells
7. One-cell thick blood vessels are known as :
(A) Alveoli
(B) Capillaries
(C) Arteries
(D) Veins
8. Observe the given figures A and B. When chhui-mui (sensitive) plant is touched, its leaves fold. This is due to:

(A) Hormonal effect
(B) Thermal effect
(C) Change in amount of water in cells
(D) Electromagnetic effect
9. Bryophyllum produces new plant through :
(A) Apical buds formed on the tip of the plant
(B) Vegetative buds produced in the notches of the leaf
(C) Flowers produced in the notches of the branches
(D) Fruits formed on the branches of the plant
10. The number of chromosomes in a cell division is halved. This kind of cell division is observed in :
(A) Only testis
(B) Only ovary
(C) Ovary and testis both
(D) All cells of the body
11. If the absolute refractive indices of two media X and Y are 6/5 and 4/3 respectively, then the refractive index of Y with respect to X will be :
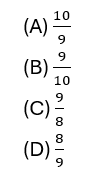
12. An object is placed at a distance of 30 cm from the pole of a concave mirror. If its real and inverted image is formed at 60 cm in front of the mirror, the focal length of the mirror is :
(A) –15 cm
(B) –20 cm
(C) +20 cm
(D) +15 cm
13. In the given figure the angle of incidence and the angle of deviation respectively are:
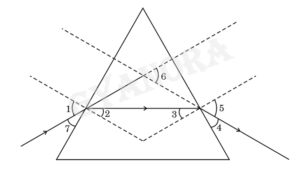
(A) 1 and 5
(B) 7 and 6
(C) 7 and 4
(D) 1 and 6
14. An electric bulb is connected to a power supply of 220 V. If the current drawn by the bulb from the supply is 500 mA, the power of the bulb is :
(A) 11 W
(B) 110 W
(C) 220 W
(D) 1100 W
15. Four identical resistors of 12 Ω each are connected in series to form a square ABCD as shown in the figure. The resistance of the network between the two points 1 and 2 is:
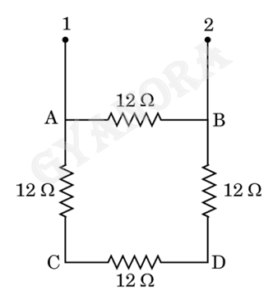
(A) 48 Ω
(B) 36 Ω
(C) 9 Ω
(D) 6 Ω
16. Identify from the following a group containing all non-biodegradable substances.
(A) Leather, Glass, Plastic
(B) Cotton, Wood, Nylon
(C) DDT, Polyester, Glass
(D) Leather, Silk, Wool
For Questions number 17 to 20,
Two statements are given — one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (A), (B), (C) and (D) as given below.
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
17. Assertion (A) : The metals high up in the reactivity series cannot be obtained from their compounds by heating with carbon.
Reason (R) : Displacement reactions can also be used to obtain metal.
18. Assertion (A): A mango seed will germinate to form a mango tree.
Reason (R): Heredity determines the process by which traits and characteristics are reliably inherited from parents to offspring.
19. Assertion (A): Nichrome is an alloy which is commonly used in electrical heating devices such as electric irons, toasters, etc.
Reason (R): The resistivity of nichrome is high and its resistance decreases with increase in temperature.
20. Assertion (A): Animals will not get energy if they eat (consume) coal as food.
Reason (R): Specific enzymes are needed for the breakdown of a particular food.
ANSWERS OF SECTION A
- (A)
- (B)
- (C)
- (A)
- (B)
- (D)
- (C)
- (C)
- (B)
- (C)
- (A)
- (B)
- (A)
- (B)
- (D)
- (C)
- (A)
- (D)
- (A)
- (C)
SECTION B (6 x 2 = 12)
Questions no. 21 to 26 are Very Short Answer Type questions.
21.
(a) In common practice silver is recovered from silver nitrate solution by the use of copper metal. Name the type of reaction that takes place in this process and give the chemical equation of the reaction involved.
(b) Name the method used for refining silver.
22.
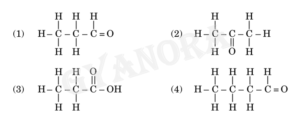
(a) Select from the following the members of same homologous series:
(b) What happens to
(i) the melting point, and
(ii) the solubility of compounds as the molecular mass of the compounds in a homologous series increases ?
23.
Name the parts of hind-brain of the human brain. Which part of the hind-brain controls involuntary actions such as blood pressure and salivation ?
24.
(a) Write one function each of the following, in a seed :
(i) Seed coat
(ii) Cotyledon
(iii) Radicle
(iv) Plumule
OR
(b) Write the main steps to culture yeast in the laboratory.
25.
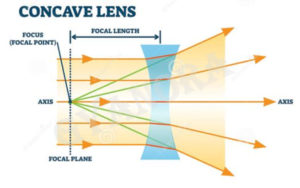
(a) Out of the two lenses, one concave and the other convex, state which one will diverge a parallel beam of light falling on it. Draw a ray diagram to show the principal focus of the lens.
OR
(b) A ray of light after refraction from a convex lens emerges parallel to its principal axis.
(i) Draw a labelled ray diagram to show it.
(ii) In this case, the incident ray before refraction from the lens passes through a point on its principal axis. Name the point.
26.
An electric kettle is rated 750 W; 220 V. Can this kettle be used in a circuit which has a fuse of current rating 3 A ? Give reason for your answer.
ANSWERS OF SECTION B
Q21.
(a)
The reaction in which copper displaces silver from silver nitrate solution is called a displacement reaction. In this reaction, copper being more reactive replaces silver from its compound.
Chemical equation:
Cu + 2AgNO₃ → Cu(NO₃)₂ + 2Ag
(b)
The method used for refining silver is electrolytic refining.
Q22.
(a)
Compounds (1) and (4) belong to the same homologous series because both are aldehydes and have the same functional group (–CHO).
(b)
(i) As the molecular mass increases in a homologous series, the melting point increases.
(ii) The solubility in water decreases with increase in molecular mass.
Q23.
The hind-brain consists of cerebellum, pons and medulla oblongata.
The medulla oblongata controls involuntary actions such as blood pressure, breathing and salivation.
Q24.
(a)
- Seed coat: Protects the seed from damage
- Cotyledon: Stores food for the embryo
- Radicle: Develops into root
- Plumule: Develops into shoot
OR
(b)
Yeast is cultured by preparing a sugar solution, adding yeast to it and keeping the mixture in warm conditions so that yeast grows and multiplies.
Q25.
(a)
A concave lens diverges a parallel beam of light falling on it.
OR
(b)
(i) A labelled ray diagram is drawn.
(ii) The incident ray passes through the principal focus of the convex lens.
Q26.
The current drawn by the kettle is
I = P/V = 750/220 ≈ 3.4 A.
Since this current is greater than 3 A, the kettle cannot be used as the fuse will melt.
SECTION C (7 x 3 = 21)
Questions no. 27 to 33 are Short Answer Type questions.
27.
(a)
(i) Define the term decomposition reaction. Write one chemical equation each for decomposition reaction where energy is supplied in the form of heat, light or electricity.
(ii) Decomposition of vegetable matter into compost is considered an exothermic reaction. Why ?
OR
(b) Why are decomposition reactions called the opposite of combination reactions ? Write one chemical equation each for these two types of reactions mentioning the name of the reactant(s) and the product(s) involved in the reactions.
28.
Write the electron-dot structures of
(i) sodium, and
(ii) oxygen.
Using these structures, show the formation of sodium oxide. Mark the anion and cation present in this compound.
(At. No. – Sodium = 11 and Oxygen = 8)
29.
(a) Define hormone.
(b) “Hormones should be secreted in precise quantities. We have a feedback mechanism through which this is done.” With the help of an example justify the statement.
30.
The lowest part of the ear called earlobe, is closely attached to the side of the head in some of us (Figure ‘X’), and not in others, called free earlobe (Figure ‘Y’). Attached and free earlobes are two variants found in human populations. The gene for free earlobe is dominant over attached earlobes.
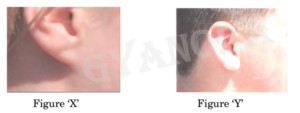
(a) A man with attached earlobes marries a woman having free earlobes. 50% of their children have free earlobes and 50% have attached earlobes. Explain the inheritance of this trait and write the trait combinations of the progeny.
(b) Write the gene combinations of the father and the mother in the above case.
31.
A convex lens forms an 8.0 cm long image of a 2.0 cm long object which is kept at a distance of 6.0 cm from the optical centre of the lens. If the object and the image are on the same side of the lens, find
(i) the nature of the image,
(ii) the position of the image, and
(iii) the focal length of the lens.
32.
Three resistors of 2 Ω, 3 Ω and 6 Ω are connected in
(i) series, and
(ii) parallel.
Draw the arrangements of the resistors and find the equivalent resistance of each arrangement.
33.
Write the essential function performed by ozone at the higher levels of the atmosphere. How is it formed in the upper atmosphere ? Write the name of the group of chemicals mainly responsible for the depletion of ozone layer.
ANSWERS OF SECTION C
Q27 (a)
A decomposition reaction is a chemical reaction in which a single compound breaks down into two or more simpler substances on supplying energy. Depending upon the type of energy supplied, decomposition reactions are of different types.
When calcium carbonate is heated, it decomposes into calcium oxide and carbon dioxide.
CaCO₃ → CaO + CO₂
Silver chloride decomposes in the presence of sunlight to form silver and chlorine gas.
2AgCl → 2Ag + Cl₂
Water decomposes into hydrogen and oxygen when electricity is passed through it.
2H₂O → 2H₂ + O₂
(ii) Decomposition of vegetable matter into compost releases heat during the process, therefore it is considered an exothermic reaction.
Q27 (b)
Decomposition reactions are called the opposite of combination reactions because in decomposition a single compound breaks into simpler substances, whereas in combination two or more substances combine to form a single compound.
In a combination reaction, calcium oxide reacts with water to form calcium hydroxide.
CaO + H₂O → Ca(OH)₂
In a decomposition reaction, calcium carbonate breaks down on heating to form calcium oxide and carbon dioxide.
CaCO₃ → CaO + CO₂
Thus, the nature of reactants and products is opposite in both reactions.
Q28
(i) Sodium has one electron in its outermost shell, while oxygen has six valence electrons. Each sodium atom loses one electron to attain a stable configuration, forming sodium ions (Na⁺). Oxygen gains two electrons to complete its octet and forms an oxide ion (O²⁻).
(ii) Two sodium atoms transfer their electrons to one oxygen atom. The oppositely charged ions attract each other due to electrostatic force and form an ionic compound called sodium oxide (Na₂O).
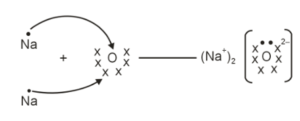
Here, sodium acts as a cation (Na⁺) and oxygen acts as an anion (O²⁻).
Q29
(a) Hormones are chemical messengers secreted by endocrine glands directly into the bloodstream. They regulate growth, development and various metabolic activities of the body.
(b) Hormones must be secreted in precise quantities because excess or deficiency can disturb body functions. This is controlled by a feedback mechanism. For example, when thyroxine hormone increases in blood, the pituitary gland reduces the secretion of TSH, thereby maintaining hormonal balance in the body.
Q30
(a) Free earlobe is a dominant trait while attached earlobe is recessive. When a man with attached earlobes marries a woman with free earlobes, the woman must be heterozygous. The genetic cross results in 50% offspring with free earlobes and 50% with attached earlobes.
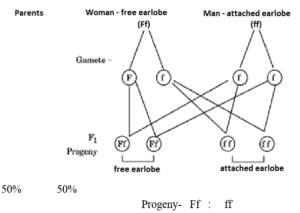
(b) The gene combination of the father is ff and that of the mother is Ff.
Q31
The magnification produced by the lens is the ratio of image height to object height.
m = 8 / 2 = 4
Since the image and object are on the same side of the lens, the image is virtual and erect.
Using magnification formula, image distance is 24 cm.
Using lens formula, the focal length of the lens is calculated to be 8 cm.
Q32
When resistors are connected in series, their resistances add up directly. Hence, equivalent resistance is
2 Ω + 3 Ω + 6 Ω = 11 Ω
In parallel combination, the reciprocal of equivalent resistance is equal to the sum of reciprocals of individual resistances. After calculation, equivalent resistance comes out to be 1 Ω.
Q33
Ozone plays a very important role in protecting life on Earth by absorbing harmful ultraviolet (UV) radiations coming from the sun. These radiations can cause skin cancer and eye damage.
Ozone is formed in the upper atmosphere when UV rays split oxygen molecules into oxygen atoms, which then combine with oxygen molecules to form ozone.
Chlorofluorocarbons (CFCs) are mainly responsible for depletion of the ozone layer.
SECTION D (3 x 5 = 15)
Questions no. 34 to 36 are Long Answer Type questions.
34.
(a)
(i) A compound ‘X’ having two carbon atoms in its molecule turns blue litmus red and 5–8% solution of ‘X’ in water is widely used as a preservative. Identify the compound ‘X’ and write its structure.
(ii) Compare its pH nature with a mineral acid.
(iii) ‘X’ on reacting with alcohols produces sweet smelling compounds, used in making perfumes. Name the reaction and write its chemical equation.
(iv) When sodium carbonate is added to ‘X’, a colourless gas is produced which turns lime water milky. Write the chemical equation for the reaction giving the name of the salt produced.
OR
(b)
(i) Differentiate between saturated and unsaturated hydrocarbons by giving one example each, with a structural formula.
(ii) Write the method of converting an unsaturated hydrocarbon into a saturated hydrocarbon. Name the industry where this reaction is commonly used.
(iii) Write the name and structure of a hydrocarbon having double bond and four carbon atoms in its one molecule.
35.
(a)
(i) “The length of the small intestine in various animals depends on the food they eat.” Justify the statement.
(ii) Discuss the role of the pancreas and bile juice in the digestion of food in human beings.
(iii) How is the small intestine designed to absorb digested food ?
OR
(b)
(i) State the role of rings of cartilage present in the throat.
(ii) Discuss the role of the ribs and diaphragm when air is taken in during the breathing cycle.
(iii) Why do we get muscle cramps during heavy exercise ? Explain.
36.
(a) The given figure shows the current passing through the straight conductor XY.
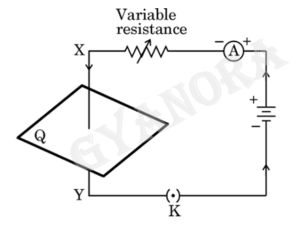
(i) Copy the diagram and draw the magnetic field lines when current flows from conductor X to Y.
(ii) Name and state the rule used in determining the direction of the magnetic field lines in the situation given above.
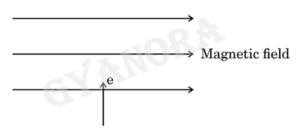
(iii) State Fleming’s left hand rule. Using this rule, determine the direction of force applied on an electron entering a uniform magnetic field as shown in the figure.
OR
(b)
(i) Define the term solenoid. Draw the pattern of the magnetic field lines in and around a current carrying straight solenoid. Mark on the pattern the
(1) direction of current,
(2) direction of field lines near the ends of the solenoid, and
(3) region where the magnetic field is uniform.
(ii) How would you make an electromagnet using a current carrying solenoid ?
ANSWERS OF SECTION D
Q34 (a)
(i) The compound ‘X’ is ethanoic acid (CH₃COOH). It contains two carbon atoms and turns blue litmus red, showing acidic nature. A 5–8% solution of ethanoic acid in water is commonly known as vinegar and is used as a preservative.
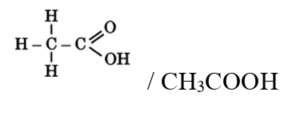
(ii)Ethanoic acid is weaker than mineral acids like hydrochloric acid because it partially dissociates in water.
(iii) When ethanoic acid reacts with alcohol in the presence of concentrated sulphuric acid, it forms sweet-smelling esters. This reaction is called esterification.
CH₃COOH + C₂H₅OH → CH₃COOC₂H₅ + H₂O
(iv) When sodium carbonate reacts with ethanoic acid, carbon dioxide gas is evolved which turns lime water milky. Sodium ethanoate is formed as the salt.
OR
Q34 (b)
(i)
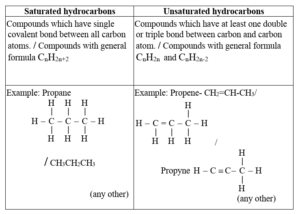
(ii) Method of converting an unsaturated hydrocarbon into a saturated hydrocarbon:
- The conversion is done by hydrogenation.
- In this process, hydrogen gas (H₂) is added to an unsaturated hydrocarbon in the presence of a catalyst like nickel (Ni).
- The double bond breaks and changes into a single bond, forming a saturated hydrocarbon.
Example reaction:
Ethene + Hydrogen → Ethane
C₂H₄ + H₂ → C₂H₆
Industry where this reaction is commonly used:
- Vegetable oil industry, where unsaturated vegetable oils are converted into saturated fats like vanaspati ghee or margarine.
(iii) Name and structure of a hydrocarbon having a double bond and four carbon atoms:
- Name: Butene (C₄H₈)
Structural formula (But-1-ene):
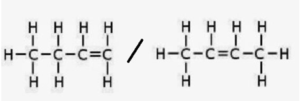
This hydrocarbon contains one double bond and four carbon atoms in one molecule.
Q35 (a)
(i) The length of the small intestine depends on the type of food an animal eats. Herbivores eat cellulose-rich food which is difficult to digest, so they have a longer small intestine. Carnivores eat meat which is easier to digest and hence have a shorter intestine.
(ii) The pancreas secretes digestive enzymes like amylase, trypsin and lipase, which help in digestion of carbohydrates, proteins and fats respectively. Bile juice emulsifies fats and makes digestion easier.
(iii) The small intestine has numerous finger-like projections called villi which increase the surface area for absorption of digested food.
OR
Q35 (b)
(i) Role of rings of cartilage present in the throat:
- The rings of cartilage are present in the trachea (windpipe).
- Their main role is to prevent the trachea from collapsing during breathing.
- They keep the airway open at all times, allowing free passage of air to and from the lungs.
(ii) Role of ribs and diaphragm during inhalation (breathing in):
- During inhalation, the external intercostal muscles contract, causing the ribs to move upward and outward.
- At the same time, the diaphragm contracts and flattens (moves downward).
- These movements increase the volume of the chest cavity, which reduces the air pressure inside the lungs.
- As a result, air from outside rushes into the lungs.
(iii) Reason for muscle cramps during heavy exercise:
- During heavy exercise, the supply of oxygen to muscle cells becomes insufficient.
- Due to lack of oxygen, muscles carry out anaerobic respiration, producing lactic acid.
- The accumulation of lactic acid in muscles causes pain and cramps.
- Cramps usually disappear after rest, as extra oxygen helps to break down lactic acid.
Q36 (a)
(i)
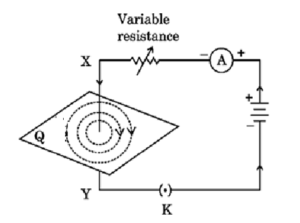
(ii) Right hand thumb rule
Statement of the rule – Imagine holding a current carrying straight conductor in the right hand such that the thumb points towards the direction of current, then the fingers will wrap around the conductor in the direction of the field lines of the magnetic field.
(iii)
- According to Fleming’s left-hand rule, stretch the thumb, forefinger and middle finger of your left hand such that they are mutually perpendicular. If the first finger points in the direction of magnetic field and the second finger in the direction of current, then the thumb will point in the direction of motion or the force acting on the conductor.
- Out of the plane/ upwards
OR
Q36 (b)
(i) A solenoid is a long cylindrical coil of insulated copper wire with many turns. When electric current passes through it, it behaves like a bar magnet.
The magnetic field inside a solenoid is strong and uniform, while outside it is weak. The direction of magnetic field lines is determined using the right-hand thumb rule.
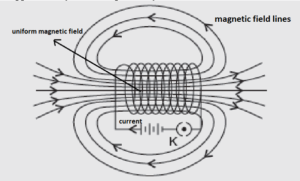
(ii) An electromagnet can be made by inserting a soft iron core inside a current-carrying solenoid. When current flows, the iron core becomes magnetised, producing a strong temporary magnet.
SECTION E (4 x 3 = 12)
The following questions are source-based / case-based questions. Read the case carefully and answer the questions that follow.
37.
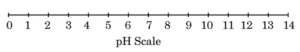
Acid-base indicators can be used to distinguish between an acid and a base. Universal indicator, a mixture of several indicators, shows different colours at different concentrations of acids and bases, thereby indicating their pH on the pH scale of 0 – 14. The pH of a solution is measured by pH paper, which is a paper impregnated with a universal indicator.
Answer the following questions :
(a) Solution P is a strong acid while solution Q is a strong base. On the pH scale, where would you place the solutions P and Q ? (1)
(b) A solution has a pH of 7. Name a compound you would use to (1)
(i) increase its pH, and
(ii) decrease its pH.
(c)
(i) When the pH of a solution is decreased from 4 to 2, what effect does it produce on its hydronium ion concentration ? State the colour change shown by the pH paper. (2)
OR
(c)
(ii) A person is feeling pain and irritation in the stomach due to indigestion. What could be the pH of the fluid in the stomach ? Write the common name of the medicines people use for remedy. Give the chemical name of “milk of magnesia” often used for this purpose. (2)
38.
When a girl is born, the ovaries already contain thousands of immature eggs. On reaching puberty, some of these start maturing. One matured egg is released every month by one of the ovaries. The two oviducts unite into an elastic bag-like structure known as uterus.
(a) Write the site of fertilization in human female. (1)
(b) How does the uterus prepare itself to receive and nurture the growing embryo ? Explain. (1)
(c)
(i) What happens when the egg is not fertilized ? (2)
OR
(c)
(ii) How does the developing embryo get nutrition from the mother’s blood ? Explain. (2)
39.
A person allowed a narrow beam of white light from the sun to enter a dark room through a small aperture and placed a glass prism in its path in such a manner that the beam falls on the face AB of the prism as shown in the figure.
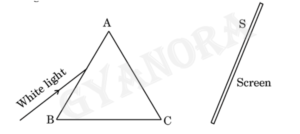
A screen S is placed on the other side of the prism, facing AC. On turning the prism slowly, a beautiful band of colours is obtained on the screen. It is the spectrum of sunlight.
(a) Name the phenomenon due to which a prism splits the incident white light into a band of colours. (1)
(b) State the reason of getting a band of seven colours in the above case. (1)
(c)
(i) Explain with the help of a labelled ray diagram, an experimental arrangement to show the recombination of the spectrum of white light. (2)
OR
(c)
(ii) Draw a labelled ray diagram to show the formation of a rainbow. (2)
ANSWERS OF SECTION E
Q37
(a) Solution P being a strong acid lies near pH 1, while solution Q being a strong base lies near pH 14 on the pH scale.
(b) To increase the pH of a neutral solution, a base like sodium hydroxide can be added, whereas to decrease pH, an acid like hydrochloric acid is used.
(c) When pH decreases from 4 to 2, the hydronium ion concentration increases hundred times. The colour of pH paper changes towards red.
Q38
(a) Fertilization in human females occurs in the oviduct (fallopian tube).
(b) The uterus prepares itself for pregnancy by thickening its inner lining and increasing blood supply to nourish the embryo.
(c) If fertilization does not occur, the uterine lining breaks down and is shed in the form of menstrual flow.
OR
(c) The developing embryo receives nutrition from the mother through the placenta, which allows exchange of nutrients and oxygen.
Q39
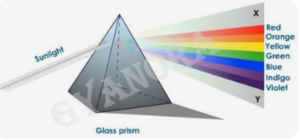
(a) A prism splits white light into seven colours due to the phenomenon of dispersion of light.
(b) Different colours have different wavelengths and hence bend by different amounts while passing through the prism.
(c ) The spectrum obtained contains seven colours because each colour travels with a different speed in glass.
OR
(c)
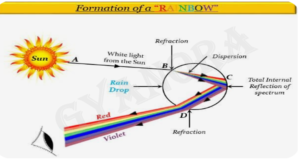
x——————x—————–x——————x
Revise your Science Concepts for Board Exam by CLICK HERE or visit Gyanora.in
Practice Chapter-Wise Extra Questions of Science for Your Board Exams by CLICK HERE
OR Explore other Chapter’s Extra Questions:
Chapter-2 Acid, Bases, and Salt
Chapter-3 Metals and Non-Metals
Chapter-4 Carbon and it’s Compounds
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6



