2024 PYQs SET-1
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections – A, B, C, D and E.
- Section A – Questions No. 1 to 20 are Multiple Choice Questions. Each question carries 1 mark.
- Section B – Questions No. 21 to 26 are Very Short Answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C – Questions No. 27 to 33 are Short Answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D – Questions No. 34 to 36 are Long Answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E – Questions No. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
SECTION – A
Select and write the most appropriate option out of the four given options for each of the questions 1–20. There is no negative mark for incorrect response.
1. Solid Calcium oxide reacts vigorously with water to form Calcium hydroxide accompanied by the liberation of heat. From the information given above it may be concluded that this reaction
(A) is endothermic and pH of the solution formed is more than 7.
(B) is exothermic and pH of the solution formed is 7.
(C) is endothermic and pH of the solution formed is 7.
(D) is exothermic and pH of the solution formed is more than 7.
2. Juice of tamarind turns blue litmus to red. It is because of the presence of an acid called :
(A) methanoic acid
(B) acetic acid
(C) tartaric acid
(D) oxalic acid
3. Select from the following a process in which a combination reaction is involved.
(A) Black and White photography
(B) Burning of coal
(C) Burning of methane
(D) Digestion of food
4. The oxide which can react with HCl as well as KOH to give corresponding salt and water is
(A) CuO
(B) Al₂O₃
(C) Na₂O
(D) K₂O
5. Consider the following cases :
(a) CaSO₄ + Al →
(b) CuSO₄ + Ca →
(c) FeSO₄ + Cu →
(d) ZnSO₄ + Mg →
The cases in which new products will form are
(A) (a) and (b)
(B) (b) and (c)
(C) (c) and (d)
(D) (b) and (d)
6. Identify the correct statement about the following reaction :
2H₂S + SO₂ → 2H₂O + 3S
(A) H₂S is oxidising agent and SO₂ is reducing agent.
(B) H₂S is reduced to sulphur.
(C) SO₂ is oxidising agent and H₂S is reducing agent.
(D) SO₂ is oxidised to sulphur.
7. Consider the following statements about homologous series of carbon compounds :
(a) All succeeding members differ by –CH₂ unit.
(b) Melting point and boiling point increase with increasing molecular mass.
(c) The difference in molecular masses between two successive members is 16 u.
(d) C₂H₄ and C₃H₆ are NOT the successive members of alkyne series.
The correct statements are –
(A) (a) and (b)
(B) (b) and (c)
(C) (a) and (c)
(D) (c) and (d)
8. Which of the following statement(s) is (are) true about human heart ?
(a) Right atrium receives oxygenated blood from lungs through pulmonary artery.
(b) Left atrium transfers oxygenated blood to left ventricle which sends it to various parts of the body.
(c) Right atrium receives deoxygenated blood through vena cava from upper and lower body.
(d) Left atrium transfers oxygenated blood to aorta which sends it to different parts of the body.
(A) (a)
(B) (a) and (d)
(C) (b) and (c)
(D) (b) and (d)
9. Select out of the following a gland which does NOT occur as a pair in the human body:
(A) Pituitary
(B) Ovary
(C) Testis
(D) Adrenal
10. In human respiratory system, when a person breathes in, the position of ribs and diaphragm will be
(A) lifted ribs and curved down diaphragm.
(B) lifted ribs and flattened diaphragm.
(C) relaxed ribs and flattened diaphragm.
(D) relaxed ribs and curved down diaphragm.
11. Identify the mode of asexual reproduction in the following organism :
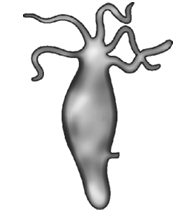
(A) Fragmentation
(B) Multiple fission
(C) Budding
(D) Binary fission
12. A cross made between two pea plants produces 50% tall and 50% short pea plants. The gene combination of the parental pea plants be
(A) Tt and Tt
(B) TT and Tt
(C) Tt and tt
(D) TT and tt
13. Consider the following statements in the context of human eye :
(a) The diameter of the eyeball is about 2.3 cm.
(b) Iris is a dark muscular diaphragm that controls the size of the pupil.
(c) Most of the refraction of the light rays entering the eye occurs at the crystalline lens.
(d) While focusing on the objects at different distances the distance between the crystalline lens and the retina is adjusted by ciliary muscles.
The correct statements are –
(A) (a) and (b)
(B) (a), (b) and (d)
(C) (a), (b) and (c)
(D) (b), (c) and (d)
14. The maximum resistance of a network of five identical resistors of each can be –
(A) 1 Ω
(B) 0.5 Ω
(C) 0.2 Ω
(D) 0.1 Ω
15. Study the I-V graph for three resistors and and select the correct statement from the following :
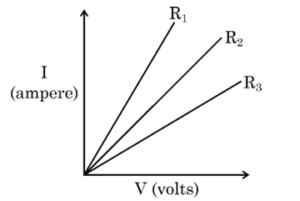
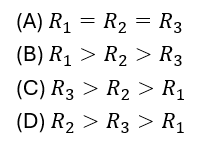
16. Strength of magnetic field produced by a current carrying solenoid DOES NOT depend upon :
(A) number of turns in the solenoid
(B) direction of the current flowing through it
(C) radius of solenoid
(D) material of core of the solenoid
Nos. 17 to 20 are Assertion – Reason based questions:
These questions consist of two statements – Assertion (A) and Reason (R).
Answer these questions selecting the appropriate option given below:
(A) Both (A) and (R) are true and (R) is the correct explanation of (A).
(B) Both (A) and (R) are true, but (R) is not the correct explanation of (A).
(C) (A) is true, but (R) is false.
(D) (A) is false, but (R) is true.
17. Assertion (A) : Different metals have different reactivities with water and dilute acids.
Reason (R) : Extraction of a metal from its ore depends on its position in the reactivity series.
18. Assertion (A) : Human female has a perfect pair of sex chromosome.
Reason (R) : Sex chromosome contributed by the human male in the zygote decides the sex of a child.
19. Assertion (A) : Myopic eye cannot see distant objects distinctly.
Reason (R) : For the correction of myopia converging lenses of appropriate power are prescribed by eye specialists.
20. Assertion (A) : The deflection of a compass needle placed near a current carrying wire decreases when the magnitude of an electric current in the wire is increased.
Reason (R) : Strength of the magnetic field at a point due to a current carrying conductor increases on increasing the current in the conductor.
ANSWERS OF SECTION-A:
- (D)
- (C)
- (B)
- (B)
- (D)
- (C)
- (A)
- (C)
- (A)
- (B)
- (C)
- (C)
- (A)
- (A)
- (C)
- (B)
- (B)
- (B)
- (C)
- (D)
SECTION – B (6 x 3 = 18)
Question No. 21 to 26 are very short answer questions.
21.
(a) “No precipitation reaction can occur without exchange of ions between the two reactants.” Justify this statement giving a balanced chemical equation for the reaction.
OR
(b) Giving one example of each, differentiate between a displacement reaction and a double displacement reaction.
22.
Photosynthesis takes place in the leaves and the food prepared by it reaches other parts of the plants. Name the process involved and explain it.
23. “Stability of DNA in a species is ensured during sexual reproduction.” Justify the statement.
24.
(a) State two laws of refraction of light.
OR
(b) Define the term absolute refractive index of a medium. A ray of light enters from vacuum to glass of absolute refractive index 1.5. Find the speed of light in glass. The speed of light in vacuum is 3 × 10⁸ m/s.
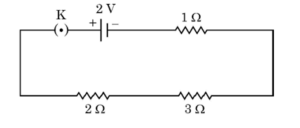
25. Use Ohm’s law to determine the potential difference across the 3 Ω resistor in the circuit shown in the following diagram when key is closed :
26. Name the term used for the materials which cannot be broken down by biological processes. Give two ways by which they harm various components of an ecosystem.
ANSWERS OF SECTION – B
21. (a)
A precipitation reaction is a type of reaction in which two aqueous solutions react with each other and exchange ions to form an insoluble solid called precipitate. Without exchange of ions, no new compound can be formed and hence precipitation does not occur. Therefore, ion exchange is necessary for a precipitation reaction.
Example:
Na₂SO₄(aq) + BaCl₂(aq) → BaSO₄(s)↓ + 2NaCl(aq)
OR
21. (b)
A displacement reaction is a reaction in which a more reactive element displaces a less reactive element from its compound.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
A double displacement reaction is a reaction in which two compounds exchange ions to form two new compounds.
Example:
Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
22.
The process by which food prepared in the leaves is transported to other parts of the plant is called translocation. It takes place through phloem tissues. Food is transported in dissolved form and energy is required for this process.
23.
Sexual reproduction produces variations in DNA due to fusion of gametes. These variations help the species to adapt to environmental changes while maintaining genetic stability. Hence, stability of DNA is ensured during sexual reproduction.
24. (a)
The two laws of refraction of light are:
- The incident ray, refracted ray and the normal lie in the same plane.
- The ratio of sine of angle of incidence to sine of angle of refraction is constant for a given pair of media.
OR
24. (b)
The absolute refractive index of a medium is the ratio of speed of light in vacuum to the speed of light in that medium.
n = c / v
v = c / n = (3 × 10⁸) / 1.5
v = 2 × 10⁸ m/s
25.
Total resistance = 1Ω + 2Ω + 3Ω = 6Ω
Current in circuit, I = V/R = 2/6 = 1/3 A
Potential difference across 3Ω resistor = I × R = (1/3) × 3 = 1 V
26.
Materials which cannot be broken down by biological processes are called non-biodegradable materials. They cause environmental pollution and ensure biomagnification in food chains, harming living organisms.
SECTION – C (7 x 3 = 21)
Question No. 27 to 33 are short answer questions.
27. It is observed that Calcium on reaction with water floats on its surface. Explain why it happens. Also write a balanced chemical equation for the reaction that occurs. What happens when the aqueous solution of the product of this reaction reacts with Carbon dioxide gas ? Write balanced chemical equation for this reaction.
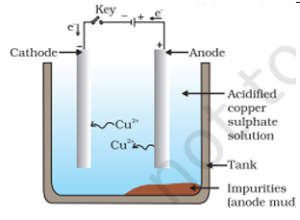
28. Draw a labelled diagram to show electrolytic refining of copper. State what happens when electric current is passed through the electrolyte taken in this case.
29.
(a) Give reasons for the following :
(i) Alveoli in lungs are richly supplied with blood capillaries.
(ii) Respiration pigment in the blood takes up oxygen and not carbon dioxide.
(iii) During anaerobic respiration, a 3-carbon molecule is formed as an end product instead of CO₂ in human beings.
OR
(b) Name the movements that occur all along the gut in human digestive system. How do they help in digestion ?
(i) Where is bile juice stored in human body ? List two roles of bile juice.
30. Explain the events that take place on a sperm reaches the oviduct till it becomes a foetus. Write the role of placenta in pregnancy.
31.
(a) Define the term power of accommodation of human eye. Write the name of the part of eye which plays a major role in the process of accommodation and explain what happens when human eye focuses
(i) nearby objects and (ii) distant objects.
OR
(b) Draw a ray diagram to show the formation of a rainbow in the sky. On this diagram mark A — where dispersion of light occurs, B — where internal reflection of light occurs and C — where refraction of light occurs. List two necessary conditions to observe a rainbow.
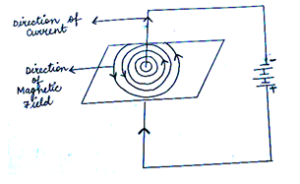
32. Draw a diagram to show the pattern of magnetic field lines on a horizontal sheet of paper due to a straight conductor passing through it at centre and carrying current vertically upwards. Mark on it
(i) the direction of current in the conductor and
(ii) the corresponding magnetic field lines. State right hand thumb rule and check whether the directions marked by you in the diagram are correct or not.
33. Use of pesticides to protect our crops affect organisms at various trophic levels especially human beings. Name the phenomenon involved and explain how does it happen.
ANSWERS OF SECTION – C
27.
Calcium floats on water because hydrogen gas bubbles produced during the reaction stick to its surface.
Ca + 2H₂O → Ca(OH)₂ + H₂
When calcium hydroxide reacts with carbon dioxide, calcium carbonate is formed.
Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
28.
In electrolytic refining of copper, impure copper is taken as anode and pure copper as cathode. On passing electric current, pure copper from the electrolyte gets deposited on the cathode, while impurities settle at the bottom as anode mud.
29. (a)
(i) Alveoli provide large surface area for efficient exchange of gases.
(ii) Haemoglobin has greater affinity for oxygen than carbon dioxide.
(iii) In anaerobic respiration, glucose breaks into lactic acid instead of carbon dioxide.
OR
29. (b)
(i) Peristaltic movements are rhythmic contractions of muscles in the alimentary canal. They help in pushing food forward and mixing it with digestive juices.
(ii) Bile juice is stored in gall bladder. It emulsifies fats and neutralises acids.
30.
After fertilisation, the zygote divides repeatedly to form an embryo which gets implanted in the uterus and develops into a foetus. Placenta provides nutrients and oxygen to the foetus and removes waste products.
31. (a)
Power of accommodation is the ability of the human eye to focus objects at different distances. Ciliary muscles help in changing the focal length of the eye lens. For nearby objects, the lens becomes thicker and for distant objects it becomes thinner.
OR
31. (b)
A rainbow is formed due to dispersion, refraction and internal reflection of sunlight in water droplets.
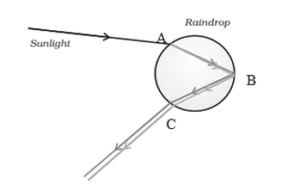
Conditions required:
- Sun should be behind the observer.
- Water droplets must be present in the atmosphere.
32.
Magnetic field lines around a current-carrying conductor form concentric circles. According to right-hand thumb rule, the thumb shows the direction of current and curled fingers show the direction of magnetic field.
33.
The phenomenon involved is biomagnification. Harmful chemicals like pesticides enter food chains and their concentration increases at higher trophic levels, affecting human beings the most.
SECTION – D (3 x 5 = 15)
Question No. 34 to 36 are long answer questions.
34.
(a) (i) Give reason why carbon cannot form C⁴⁺ cations nor C⁴⁻ anions but can form covalent compounds.
(ii) What is homologous series of carbon compounds ? Write the molecular formula of any two consecutive members of homologous series of aldehydes.
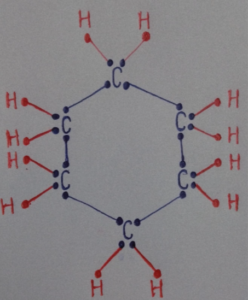
(iii) Draw the structure of the molecule of cyclohexane (C₆H₁₂).
OR
(b) (i) Name a commercial important carbon compound having a functional group –OH and write its molecular formula.
(ii) Write chemical equation to show its reaction with :
(1) Sodium metal
(2) Excess conc. sulphuric acid
(3) Ethanoic acid in the presence of an acid catalyst
(4) Acidified potassium dichromate
Also write the name of the product formed in each case.
35.
(a) (i) Distinguish between hormonal co-ordination in plants and animals.
(ii) Which part of the brain is responsible for –
(1) intelligence
(2) riding a bicycle
(3) vomiting
(4) controlling hunger
(iii) How is brain and spinal-cord protected against mechanical injuries ?
OR
(b) (i) What are tropic movements ? Give an example of a plant hormone which inhibits growth and (2) promotes cell division.
(ii) Explain directional movement of a tendril in pea plant in response to touch. Name the hormone responsible for this movement.
36.
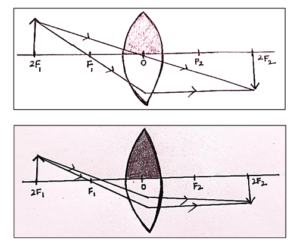
(a) Upper half of a convex lens is covered with a black paper. Draw a ray diagram to show the formation of image of an object placed at a distance of 2F from such a lens. Mention the position and nature of the image formed. State the observable difference in the image obtained if the lens is uncovered. Give reason to justify your answer.
(b) An object is placed at a distance of 30 cm from the optical centre of a concave lens of focal length 15 cm. Use lens formula to determine the distance of the image from the optical centre of the lens.
ANSWERS OF SECTION – D
34. (a)
(i) Carbon does not form C⁴⁺ or C⁴⁻ ions due to high ionisation energy. Hence, it forms covalent bonds.
(ii) A homologous series is a group of compounds having the same functional group and similar chemical properties.
Examples: Ethanal (C₂H₄O) and Propanal (C₃H₆O).
(iii) Cyclohexane consists of six carbon atoms arranged in a ring.
OR
34. (b)
(i) Ethanol is a commercially important compound having –OH functional group. Molecular formula is C₂H₅OH.
(ii) Reactions:
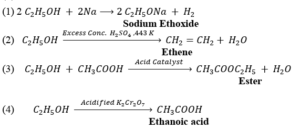
35. (a)
(i) Hormonal coordination in plants is slow and chemical, while in animals it is fast and controlled by nervous system.
(ii)
(1) Cerebrum controls intelligence
(2) cerebellum controls balance
(3) medulla controls vomiting
(4) hypothalamus controls hunger.
(iii) Brain and spinal cord are protected by skull and vertebral column.
OR
35. (b)
(i) Tropic movements are directional growth movements of plants.
(1) Growth inhibitor hormone: Abscisic acid
(2) Cell division promoter: Cytokinin
(ii) Tendril movement in pea plant occurs due to auxin and is called thigmotropism.
36.
(a) Even when half of a convex lens is covered, a complete image is formed but with reduced brightness.
(b) Here u = – 30 cm, ƒ = –15 cm, 𝜈 = ?
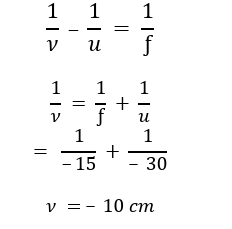
SECTION – E (3 x 4 = 12)
Question No. 37 to 39 are case based / data based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
37. Salts play a very important role in our daily life. Sodium chloride which is known as common salt is used almost in every kitchen. Baking soda is also a salt used in faster cooking as well as in baking industry. The family of salts is classified on the basis of cations and anions present in them.
(a) Identify the acid and base from which Sodium chloride is formed.
(b) Find the cation and the anion present in Calcium sulphate.
(c) “Sodium chloride and washing soda both belong to the same family of salts.” Justify this statement.
OR
(c) Define the term pH scale. Name the salt obtained by the reaction of Potassium hydroxide and Sulphuric acid and give the pH value of its aqueous solution.
38. Asexual reproduction involves a single parent to produce offsprings without the formation of gametes. It occurs by the following ways : Fission, Budding, Fragmentation, Spore formation and Regeneration. In one of the methods like regeneration, Planaria A is cut horizontally into three pieces – L, M and N and Planaria B is cut vertically into two equal halves – O and P.
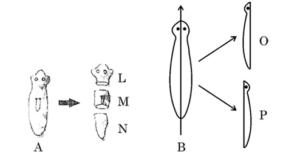
(a) Which of the pieces of the two Planaria could regenerate to form a complete organism ?
(b) Give an example of an organism which follows the same mode of reproduction as Planaria.
(c) What is the meaning of ‘development’ in regeneration ?
(d) Differentiate between regeneration and fragmentation.
39. When electric current flows in a purely resistive circuit electrical energy gets fully converted into heat energy. The amount of heat produced (H) in the circuit is found to be directly proportional to (i) the square of current (I²), (ii) the resistance (R) of the conductor and (iii) the time (t) for which current flows. In other words H = I²Rt. Electrical devices such as an electric fuse, electric heater, electric iron etc. are all based on this effect called heating effect of electric current.
(a) List two properties of electric element.
(b) List two properties of electric fuse.
(c) Name the principle on which an electric fuse works. Explain how a fuse wire is capable of saving electrical appliances from getting damaged due to accidentally produced high currents.
OR
(c) The power of an electric heater is 1100 W. If the potential difference between the two terminals of the heater is 220 V, find the current flowing in the circuit. What will happen to an electric fuse of rating 5 A connected in this circuit ?
ANSWERS OF SECTION – E
37.
(a) Sodium chloride is formed from hydrochloric acid and sodium hydroxide.
(b) Calcium sulphate contains Ca²⁺ cation and SO₄²⁻ anion.
(c) Both sodium chloride and washing soda contain sodium ion, hence belong to same family of salts.
OR
(c) pH scale measures acidity or basicity of a solution.
Salt formed: Potassium sulphate (K₂SO₄)
pH of aqueous solution = 7
38.
(a) All pieces of Planaria can regenerate into complete organisms.
(b) Hydra reproduces by regeneration.
(c) Development refers to growth and differentiation of cells.
OR
(c) Regeneration forms a complete organism, whereas fragmentation breaks body into parts.
39.
(a) Electric element has high resistance and high melting point.
(b) Fuse wire has low melting point and thin structure.
(c) Fuse works on heating effect of electric current.
OR
(c)Current I = P/V = 1100/220 = 5 A
Fuse of rating 5 A will be safe but will melt if current exceeds this value.
x——————x—————–x——————x
Revise your Science Concepts for Board Exam by CLICK HERE or visit Gyanora.in
Practice Chapter-Wise Extra Questions of Science for Your Board Exams by CLICK HERE
OR Explore other Chapter’s Extra Questions:
Chapter-2 Acid, Bases, and Salt
Chapter-3 Metals and Non-Metals
Chapter-4 Carbon and it’s Compounds
Follow us on:
YouTube: www.youtube.com/@Gyanora
Telegram: t.me/gyanora
Instagram: https://www.instagram.com/gyan_ora?igsh=aDQxbDk1aDdleHJ6



